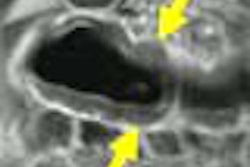
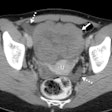
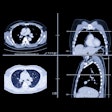
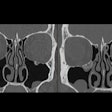
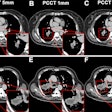
Computer-aided detection developer R2 Technology has received Food and Drug Administration clearance for its ImageChecker CT CAD software. ImageChecker CT was cleared for the detection of solid lung nodules during review of multidetector CT (MDCT) chest exams, according to the Sunnyvale, CA-based vendor.
Alexian Brothers Hospital Network in Chicago, which installed R2's ImageChecker CT lung-analysis workstation earlier this year, will be the first facility to upgrade to the newly approved software, R2 said.
By AuntMinnie.com staff writersJuly 12, 2004
Related Reading
R2's Klein resigns, interim CEO named, June 9, 2004
R2 closes U.S. government deal, May 27, 2004
MedAssets, R2 sign mammography deal, May 25, 2004
Premier, R2 ink mammography deal, April 28, 2004
R2 gets FDA OK for use with Hologic FFDM, April 14, 2004
Copyright © 2004 AuntMinnie.com