Fujifilm Medical Systems USA plans to unveil two new digital radiography (DR) products at RSNA 2015 in Chicago.
The company will show its FDR Visionary suite and FDR D-Evo GL long-view DR detector.
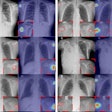
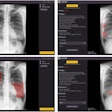
The FDR Visionary suite is a flexible, automated system that lets technologists and clinicians choose an imaging method for each exam by selecting from a range of advanced applications, including tomosynthesis, dual energy subtraction, and long-view imaging.
It also has Fuji's cesium iodide (CsI) detectors and image processing to improve positioning repeatability and workflow, image quality, and diagnostic visibility for challenging views and fine detail exams. The FDR Visionary suite is currently under 510(k) review by the U.S. Food and Drug Administration (FDA).
Meanwhile, the FDR D-Evo GL DR detector is designed to acquire long-length radiography for scoliosis imaging and/or long leg exams with a low-dose, single exposure. The 17 x 19-inch field-of-view is intended to improve image quality and dose for upright long-length radiography exams.
Fuji also announced that it has submitted the second module of its premarket approval (PMA) application to the FDA for digital breast tomosynthesis (DBT) as an optional upgrade for the Aspire Cristalle mammography system, which is known as Amulet Innovality outside of the U.S. The company plans to file the remaining modules of the PMA for DBT within the coming year.
The optional DBT upgrade has been available since May 2013 in Europe, Asia, and Latin America, Fuji said.