Medical software developer Densitas is directing attention to a recent review that validated the clinical use of its automated breast-density measurement software to help stratify patients based on their risk for breast cancer.
The findings were published online February 5 in Breast Cancer Research.
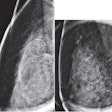
Researchers from the University of Manchester in the U.K. reviewed five different methods for assessing breast density in 366 women participating in the Predicting Risk of Cancer at Screening (PROCAS) study. They found that both the Densitas software and the VolparaDensity algorithm (Volpara Solutions) could be used practically in breast cancer risk models due to the strong association of these density measurements with the risk of breast cancer.
Visual and semiautomated density assessments also show strong relationships with breast cancer risk, but the researchers judged these approaches to be impractical in population-based screening, Densitas said.
The company also said that several scientific and poster presentations at ECR 2018 in March will further confirm the clinical validation of its breast density software.