Minnies finalists, page 3
Scientific Paper of the Year
Reading Race: AI Recognises Patient's Racial Identity In Medical Images. Imon Banerjee I, Bhimireddy A et al, arXiv.org, July 21. Learn more about this study.
The first study up for the Scientific Paper of the Year begged the question, "Is radiology artificial intelligence (AI) technology racist?"

In a preprint paper published arXiv.org, a group of researchers assembled by Dr. Judy Gichoya of Emory University in Atlanta found extremely high accuracies -- up to an area under the curve (AUC) of 0.99 -- for the ability of deep-learning models to recognize a patient's self-described racial identity from radiology images.
Here's the rub: The researchers were unable to ascertain exactly how the algorithm learns to predict racial identity.
"Every radiologist I have told about these results is absolutely flabbergasted," wrote co-author Dr. Luke Oakden-Raynor of the University of Adelaide in Australia, in a blog post.
Further investigation and research into the human-hidden but model-decipherable information that these images appear to contain related to racial identity is warranted, the authors concluded.
The study has since been formally published in Lancet Digital Health with the same conclusions.
Mandating Limits on Workload, Duty, and Speed in Radiology. Alexander R, Waite S, et al, Radiology, June 14. Learn more about this study.
The second finalist for Scientific Paper of the Year recommended hitting the brakes when it comes to regulating the workloads of radiologists in response to a rise in the U.S. of so-called "reckless reading lawsuits."

Radiology workloads have seen relentless growth in recent years, with no signs of slowing down, said a group of researchers at SUNY Downstate Medical Center in Brooklyn, NY. They asked, "Is it medical negligence to interpret an image quickly?"
In their review and analysis, the authors cited a case from 2020 that alleged a subdural hematoma in a 64-year-old man was missed because the radiologist spent a total of six minutes and 27 seconds reading a CT exam of the head and cervical spine (as determined by subpoenaed keystroke data).
The plaintiff's attorney argued that the average viewing time of each image was under a second per image, and therefore the radiologist was lax (i.e., not sufficiently careful), thus engendering a larger settlement.
The group in Brooklyn corrected this math. According to their calculations, the precise time it takes is 0.86 seconds per image, and this is in fact the current standard of care in radiology.
"Implying that such behavior is negligent fails logic," the authors wrote.
Moreover, in the past 20 years, no studies have quantified how many examinations or images radiologists can review in any period while maintaining accuracy, they concluded.
Best New Radiology Device
Naeotom Alpha, Siemens Healthineers
For years, photon-counting CT was a gleam in radiology's eye -- a technology that promised to raise CT instrumentation above the slice wars to a new level of performance. But despite promising research, the technology seemed long in coming to real-world clinical practice.
All that changed in September 2021, when the U.S. Food and Drug Administration cleared Naeotom Alpha, a photon-counting CT scanner from Siemens Healthineers that's designed for routine clinical use. The clearance arrived too late for the 2021 Minnies competition, but the glow of the news has clearly carried over into 2022, with the Minnies expert panel choosing the system to advance to the finalist round.
 The Naeotom Alpha CT scanner.
The Naeotom Alpha CT scanner.Naeotom Alpha is a dual-source CT scanner, with a 0.25-second gantry rotation time, 66-millisecond temporal resolution, and an x-ray generator with power reserve up to 1,300 mA. The system has volumetric coverage of 74 cm per second.
Siemens exhibited Naeotom Alpha for the first time at the RSNA 2021 conference, and since then has been installing the scanner at luminary sites around the world. These sites have begun producing research that highlights the many potential benefits of photon-counting CT, ranging from lower radiation dose to the ability to spot pathologies missed by conventional CT scanners.
For example, researchers from Mayo Clinic in Rochester, MN -- the first site in the U.S. where the system was installed -- touted many of the technology's benefits in a presentation in September at the International Society for Computed Tomography (ISCT) annual meeting. Another study out of Mayo, published in September in Radiology, found that photon-counting CT combined with deep learning to correct for noise improves visualization of bone disease caused by multiple myeloma.
Meanwhile, researchers from Germany demonstrated how photon-counting CT can drive down radiation dose by more than one-third, while a group from Duke University -- another early adopter of the technology -- found that it can visualize cerebrospinal fluid (CSF) venous fistula (CVF) better than traditional imaging approaches such as CT myelography or MRI
Regardless of how Naeotom Alpha fares in the next round of the Minnies, photon-counting CT looks set to have a dramatic impact on the practice of radiology around the world.
SoftVue 3D whole-breast ultrasound tomography system, Delphinus Medical Technologies
The other finalist in the Best New Radiology Device category of the Minnies also received FDA premarket approval in fall 2021: the SoftVue 3D whole-breast tomography system from Delphinus Medical Technologies.
SoftVue is designed to address a nagging problem with breast imaging -- conventional x-ray-based mammography does not perform well in women with dense breast tissue. Other modalities have been proffered as an adjunct to mammography -- like MRI and ultrasound -- but these technologies have disadvantages of their own.
 SoftVue 3D whole-breast ultrasound tomography system. Image courtesy of Delphinus Medical Technologies.
SoftVue 3D whole-breast ultrasound tomography system. Image courtesy of Delphinus Medical Technologies.With SoftVue, a woman lies prone on the system's table, with her breasts hanging through a hole in the table into a warm water bath. The breasts are stabilized and centered with a disposable gel pad, and SoftVue then scans each breast from chest wall to nipple tip in an average of three minutes. The exam is performed with a transducer and captures new images every 2 mm.
SoftVue's 360° ring-shaped ultrasound transducer incorporates over 2,000 elements using the company's TriAD triple acoustic detection technology, which gathers reflected echoes as well as quantifies the sound speed and attenuation signals as they are transmitted through the breast. The system produces tomographic coronal images every 2 mm, which breast imagers can use to distinguish normal tissue from suspicious areas.
The novelty of SoftVue is borne out by the fact that the company navigated the premarket approval (PMA) regulatory process, which is more rigorous than 510(k) clearance process received by most medical imaging devices. Delphinus conducted a clinical study in support of the PMA that found that use of SoftVue increased radiologist sensitivity by 20% and specificity by 8%, and the radiologists were able to identify up to 20% more cancers with greater accuracy than mammography alone.
The system's approval indicates that it's intended to be used as an adjunct to mammography for breast cancer screening in asymptomatic women with dense breast tissue. It can be used in the same visit as a screening mammogram, and SoftVue images are intended to be interpreted with the mammogram, according to FDA documentation.
With PMA approval in hand, Delphinus is moving to get installations in place and to sponsor research on the system. A study published in Journal of Breast Imaging in 2021 found that 95% of breast cancers detected with the system are located at the circumferential fat-glandular interface (FGI), and the location of masses relative to the FGI could be helpful in adding to computer-aided detection algorithms.
Best New Radiology Software
AI Operating System, Aidoc
Introduced at RSNA 2021 in Chicago, AI Operating System (OS) is a platform designed to provide the infrastructure to support the adoption and integration of artificial intelligence (AI) algorithms into the radiology workflow.
The vendor-agnostic "operating system" makes it easier for healthcare institutions to deploy radiology AI by orchestrating a range of AI algorithms from Aidoc as well as third-party developers, according to the vendor. AI OS enables each radiology subspecialty to make use of AI algorithms within their own workflow, Aidoc said.
 AI Operating System. Image courtesy of Aidoc.
AI Operating System. Image courtesy of Aidoc.The platform facilitates access to Aidoc's portfolio of nine algorithms cleared by the U.S. Food and Drug Administration (FDA) for triage and detection across a range of radiology applications. In addition, it provides access to AI algorithms from companies such as Icometrix, Imbio, Riverain Technologies, ScreenPoint Medical, and Subtle Medical.
AI-based image analysis is used to match the most compatible algorithm with the relevant scan. Furthermore, AI OS can also help to improve communication between caregivers in a health network by providing automated alerts and relevant information for radiologists and other physicians to review, Aidoc said.
Koios DS, Koios
The other finalist for Best New Radiology Software, Koios DS, is an ultrasound artificial intelligence (AI) platform aimed at helping radiologists improve their accuracy in detecting breast cancer (via its BreastDS algorithm) and thyroid cancer (using its ThyroidDS application), according to Koios.
A study presented at SIIM 2022 showed that BreastDS can outperform the commonly used Tyrer-Cuzick model for predicting malignancy risk on breast lesions found on breast ultrasound exams. And, in a study involving over 200 lesions found on breast ultrasound, researchers from Yale University found that BreastDS correctly classified all malignant cases and downgraded many lesions initially deemed to be suspicious.
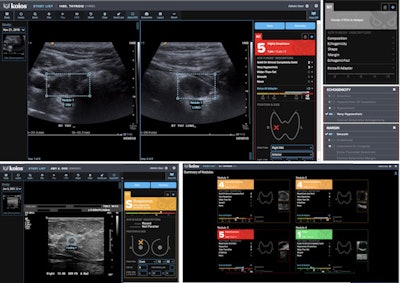 Koios-DS is an ultrasound AI platform for assessing breast and thyroid lesions. Image courtesy of Koios.
Koios-DS is an ultrasound AI platform for assessing breast and thyroid lesions. Image courtesy of Koios.Koios DS received the CE Mark for Koios DS in March 2021, as well as clearance from Health Canada in November 2021. In December 2021, Koios secured clearance from the U.S. Food and Drug Administration (FDA) in December 2021 for the use of Koios DS in both breast and thyroid ultrasound exams.
The firm, which was a partner in GE Healthcare's Edison Developer Program, also signed a deal last year to make BreastDS available on GE's Invenia ABUS 2.0 automated whole-breast ultrasound scanning system.
Best New Radiology Vendor
Annalise.ai
This artificial intelligence (AI) software developer was formed in 2019 as a joint venture between healthcare AI firm harrison.ai and Australian diagnostic imaging services provider I-MED Radiology Network.
Designed to help radiologists interpret chest x-rays, the company's first product -- Enterprise CXR -- can detect 124 findings and provides users with a confidence bar that indicates the model's estimated likelihood of the finding and its uncertainty. Enterprise CXR has received the CE Mark for use in Europe and is also available for clinical use in Australia, New Zealand, and Malaysia.
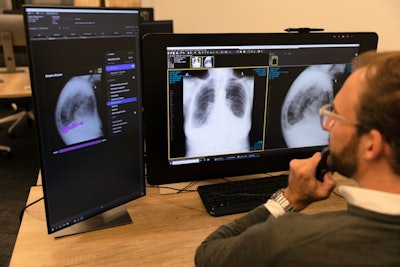 Annalise.ai workstation. Image courtesy of Annalise.ai.
Annalise.ai workstation. Image courtesy of Annalise.ai.Earlier this year, Annalise.ai received U.S. Food and Drug Administration (FDA) clearance for a part of the software -- Enterprise CXR Triage Pneumothorax -- as a tool for triage and notification of pneumothorax and tension pneumothorax on chest x-rays.
A second product -- CXR Edge -- is designed to aid clinicians in interpreting chest x-rays at the point of care. It can assess chest x-rays for up to 95 findings and notify users of suspected findings on the device in about 10 seconds, according to the company. CXR Edge is available in Australia via a distribution partnership with Fujifilm Australia.
InformAI
This artificial intelligence (AI) software firm develops applications for detecting and classifying pathology, as well as predicting patient outcomes.
On CT scans, Paranasal Classifier detects and distinguishes between conditions prevalent in the paranasal sinuses, according to InformAI. Its second classifier software -- Brain Cancer Classifier -- spots and characterizes brain cancer on MRI.
InformAI has also trained software that predicts patient outcomes associated with cardiac and thoracic surgeries. These decision tree-based models can be built to specific customer requirements and provide risk estimates for outcomes such as mortality, stroke, prolonged ventilation, infection, a repeat operation, and prolonged hospitalization, according to the company.
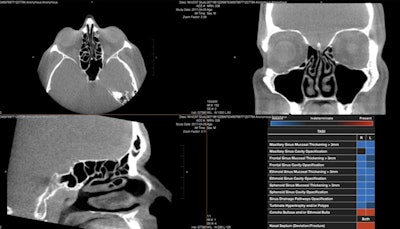 InformAI’s lead product – SinusAI – is an AI algorithm that detects and distinguishes several conditions and features prevalent in the paranasal sinuses. Image courtesy of InformAI.
InformAI’s lead product – SinusAI – is an AI algorithm that detects and distinguishes several conditions and features prevalent in the paranasal sinuses. Image courtesy of InformAI.In 2020, InformAI was awarded a $225,000 Small Business Technology Transfer grant from the National Science Foundation to build an AI application to assist organ transplant surgeons in the matching of donor organs with patient recipients.
Recently, InformAI was also invited by the University of Texas Southwestern Medical Center to join its Entrepreneur in Residence program. For the next three years, this program will connect InformAI with investment and commercialization experts, according to the firm.
Best Educational Mobile App
iRadTech, Ballinger and Bruckner (iOS, Android)
iRadTech is a radiographic positioning guide geared toward medical imaging professionals and students. Available for both Apple and Android smartphone and tablets and as a web app, iRadTech is designed to serve essentially as an x-ray "pocket guide" or reference booklet, according to its developer RadTech Resource Group.
iRadTech features detailed instructions for more than 200 radiography positions. It also includes an enlargeable photograph of a properly positioned human model demonstrating the correctly collimated area of interest with the central x-ray point, according to the developers. The app can serve as a guide and study aid for radiography students, as well as a reference and review tool for technologists and clinical instructors.
iRadTech was developed by radiologic technologists Philip Ballinger, PhD, a professor emeritus at Ohio State University's radiologic technology division, and Terri Bruckner, PhD, a longtime radiography educator.
RadDiscord, a server on Discord, Dr. Grace Zhu (iOS, Android)
Now a two-time finalist for Best Educational Mobile App, RadDiscord was launched in 2020 as a server (#RadDiscord) on the Discord platform to provide a community for radiology residents to collectively study for the radiology board exam.
 RadDiscord educational content created by radiology residents. Image courtesy of Dr. Grace Zhu.
RadDiscord educational content created by radiology residents. Image courtesy of Dr. Grace Zhu.RadDiscord's more than 3,200 members from around the world can interact in real-time, as well as access board review lectures, Anki flashcard decks, study voice channels, and text channels with case-of-the-day and subject content. The community aims to provide equal access to radiology knowledge to all practicing radiologists and trainees, as well as to continuously improve how radiologists are educated.
RadDiscord was created by Dr. Grace Zhu, a former radiology resident and now an assistant professor in the University of Utah's department of radiology and imaging sciences. For the 2022-2023 academic year, radiology resident Dr. Fatima Elahi of Chicago will be president of Discord while radiology resident Dr. Cam Henry of Vanderbilt University will serve as vice president.
Previous page | 1 | 2 | 3