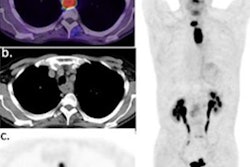
Sofie Biosciences has performed imaging on the first patient in a phase II clinical trial to test the use of its gallium-68 (Ga-68) fibroblast activation protein inhibitor (FAPI) PET radiotracer in patients with pancreatic cancer.
The procedure took place at NYU Langone Health. Langone Health and Mayo Clinic are the first two sites to participate in the study, with additional sites expected to join in the coming months.
Sofie's Ga-68 FAPI-46 tracer was developed by a team at University Hospital Heidelberg in Heidelberg, Germany, to image fibroblast activation protein (FAP), a protein that is highly expressed by fibroblast cells in tumors.