CT perfusion imaging can spot significant changes in blood flow and volume soon after administration of antiangiogenic therapy, researchers from Munich, Germany, report in a new animal study. Faster assessment can lead to nimbler, more flexible cancer care compared to traditional tumor size measurements.
The emergence of several antiangiogenic therapies for cancer demands a shift in the focus of imaging, from measuring tumor size changes with treatment to the search for surrogate markers of clinical treatment response.
In this environment, CT perfusion may be an ideal modality for assessing tumor response to antiangiogenic therapy: It's fast, it's technically easy, and it requires no special equipment, said Konstantin Nikolaou, MD, associate chair of clinical radiology at Munich University Hospitals.
Cancer is typically treated with radiation and chemotherapy, and tumors are known to shrink in size after treatment, a response traditional CT measures well, if slowly: It can take weeks of therapy to produce tumor size changes big enough to measure, and size assessment alone is only a crude measure of tumor activity. Nikolaou spoke at last month's International Society for Computed Tomography (ISCT) meeting in San Francisco.
Pharmaceutical companies want to quickly assess the efficacy of their expensive new treatment agents, which enables rapid changes in therapy as the patient's condition demands. Nikolaou said his facility is often approached by pharmaceutical companies looking for new ways to quickly measure tumor response to antiangiogenic therapies, and his group is eager to help with the task.
"We want to test them in combination with several modalities -- ultrasound, MRI, CT -- and new drugs and tumor models to evaluate if there is a response to these entities," Nikolaou said.
Angiogenesis is new blood vessel development that occurs in several processes, including atherosclerosis and tumor growth, Nikolaou said. "It is a process that you can stop at many different steps with many different therapeutic approaches, and any tumor that wants to reach a certain size needs some new vessels."
Each of the various noninvasive imaging modalities (MRI, ultrasound, CT) has its specific advantages and drawbacks, but monitoring of perfusion parameters with one or more modalities is already a clinical reality, Nikolaou said. What's needed now are studies that can optimize protocols to achieve the full potential of antiangiogenesis therapy monitoring, a task that is tailor-made for animal studies.
"The beauty of an animal model is that you can do several scans over days, and check what kind of therapies and what kinds of tumor models will give you the best time point to evaluate therapeutic response -- something you will not necessarily be able to do with patients," Nikolaou said.
The study examined the use of the antiangiogenic agent sorafenib, a small-molecule tyrosine kinase inhibitor, on 15 Copenhagen rats, seven of which were injected with prostate carcinoma MLLB2 (Mat-LyLu-B2). On day 1, the researchers acquired a baseline CT scan, followed by six days of sorafenib therapy (10 mg/kg body weight) and an additional scan each day. On day 7, a final CT scan was acquired and the tumors were excised for immunohistochemical staining and analysis.
Following administration of 2 mL/kg iopromide (370 mg iodine), CT perfusion images were acquired on a dual-source CT scanner (Somatom Definition Flash, Siemens Healthcare, Erlangen, Germany) at 200 mAs, 80 kV, and 128 x 0.6 mm.
 |
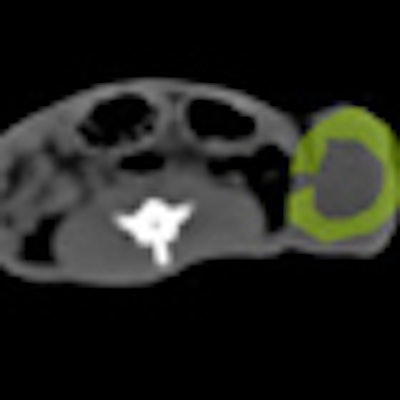
| Blood flow decreased in mice with induced prostate cancer following one week of antiangiogenic therapy (above); no change was seen in control animals (below). |
 |
Baseline scans typically show a small solid tumor, while minimal rim uptake is visible at follow-up seven days later, Nikolaou said. Discussion is ongoing as to what should be measured -- only the tumor rim, or the center? Today many different models are used; an important one is perfusion permeability.
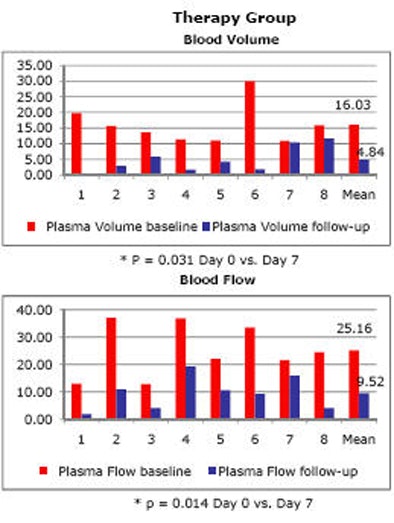
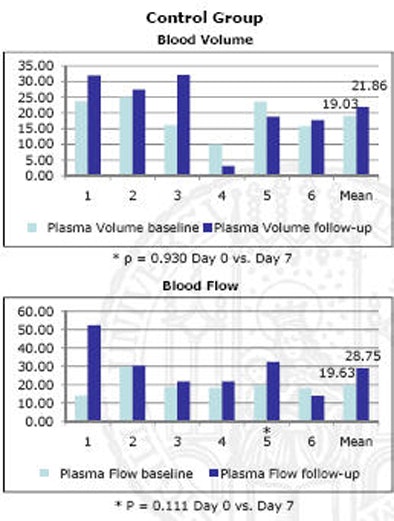
The results indicated that "every single animal showed a tumor effect in terms of blood volume and blood flow," Nikolaou said. "And you don't want to have to look at 100 patients to see the effect you want to see in every patient."
One animal with disease did not show a statistically significant decrease in blood volume over the week's treatment time, but blood flow did drop, and the rest of the diseased animals showed significant decreases in both measures, he said. In contrast, the control group showed no significant decrease in any of the animals, and some actually showed an increase in blood volume and blood flow.
 |
| Charts quantify significant drop in blood volume (top) and blood flow (bottom) following one week of sorafenib antiangiogenic therapy. |
 |
To assess the utility of antiangiogenic agents, it's important to look at the use of different imaging modalities, which can be directly compared with the use of animal models, using MRI as well as CT, he said.
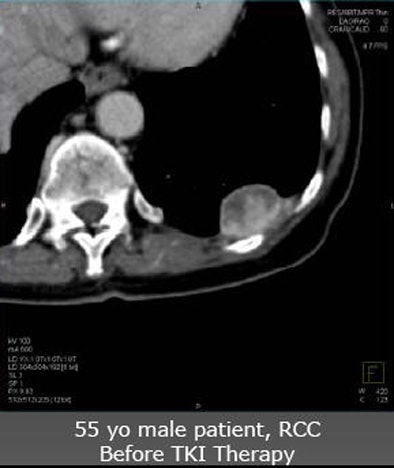 |
| Future studies will focus on clinical experience with angiogenic agents. In a patient with renal cell carcinoma with pleural metastasis (above), decreases in tumor perfusion are readily seen after three weeks of sorafenib antiangiogenic therapy (below). |
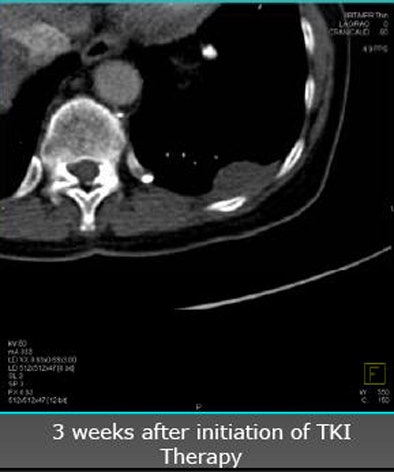 |
"It is already a clinical reality and you will see more and more patient studies -- but I think we also need studies using animal models to optimize our imaging strategies," Nikolaou said.
For example, what is the optimal scanning frequency, the best postprocessing method, the best way to quantify the results? What is the prognostic value of the different perfusion parameters? "I think you're going to see animal models looking at these different approaches," he said.
CT has several clear advantages in perfusion assessment because it's fast, and there's no need to have the latest scanner model to perform it well. "And there is reproducible quantification with the use of iodine," Nikolaou said. "But radiation is a topic, and we will need to optimize our protocols."
By Eric Barnes
AuntMinnie.com staff writer
June 14, 2010
Related Reading
Dual-energy VC aims for noninvasive polyp evaluation, June 2, 2010
CT perfusion distinguishes HCC from other liver lesions, April 29, 2010
Perfusion CT handily distinguishes malignant neck nodes, January 25, 2010
CT of colorectal liver mets may predict survival, December 1, 2009
PET/CT directs head and neck cancer treatment, November 17, 2009
Copyright © 2010 AuntMinnie.com