CHICAGO - Pulmonary CT angiography is the gold standard modality for diagnosing pulmonary embolism (PE), and detecting pulmonary perfusion defects at CT is considered good evidence for the presence of PE. But not all perfusion defects represent PE. Can dual-energy CT distinguish non-PE causes of perfusion defects from the real thing?
Researchers from Seoul National University Hospital in South Korea found that they can -- with the aid of iodine perfusion mapping in addition to dual-energy pulmonary CT angiography (CTA).
"With CT angiography alone, we cannot know the effect of embolism on pulmonary perfusion," said Dr. Mi-Jin Kang in a presentation on Monday at the RSNA meeting.
Moreover, a perfusion deficit is not necessarily a clot. "Several lesions that cause increased parenchymal attenuation or profound decreased parenchymal attenuation can also manifest as perfusion defects," she said. "These lesions are rarely confused with embolism because we are able to match it in the CT scan."
Unfortunately, some areas of perfusion defects aren't associated with parenchymal lesions and therefore can't be matched to the scans, she said. "In these cases we were concerned whether these lesions were indeed true embolisms," Kang said.
The researchers investigated the location and incidence of lung perfusion defects that were inconsistent with pulmonary embolism on color-coded perfusion maps of dual-energy pulmonary CTA.
Over a three-month period, 105 patients underwent pulmonary CT angiography. The researchers selected 43 patients (18 men, 25 women; mean age, 56.9 years) who had no evidence of PE at the initial scan, and were also confirmed to be free of PE at a two-month follow-up scan.
Comorbidities included a solid organ tumor in 18 patients, heart problems in five patients, hematologic malignancies in four patients, and pneumonia in five patients.
Among the 860 segments imaged with dual-energy CT, 122 segments that complicated the analysis of perfusion maps were excluded, leaving a total of 738 segments in 43 patients to be included in the study, Kang said. Dual-energy CT images were acquired on a Somatom Definition scanner (Siemens Healthcare, Malvern, PA) in dual-energy mode.
Acquisition parameters included tube voltages of 80 kVp (213 mAs) and 140 kVp (51 mAs) using Siemens' CARE Dose 4D tube current modulation, 1.2 to 1.4-mm collimation, and 1.5-mm reconstruction thickness.
Scanning from the lung base to the apex was initiated six seconds after a region of interest in the right ventricle reached 120 HU, following injection of 80 mL of iopromide (Ultravist 370, Bayer HealthCare Pharmaceuticals, Wayne, NJ) at 4 cc/sec, then a 10% contrast solution at 4 cc/sec for 10 seconds.
After the scan, three sets of images were automatically generated from each study, including 140 kVp images, 80 kVp images, and weighted-average images. A Siemens Leonardo workstation was used to generate iodine maps and fused images using lung PBV mode.
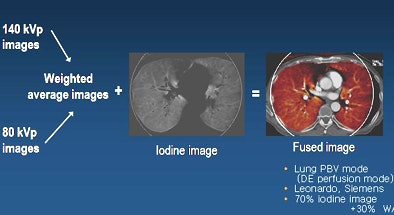 |
| After scanning, three sets of images were automatically generated: 140 kVp images, 80 kVp images, and weighted-average images. The workstation used the weighted-average images to create iodine maps and fused images using lung PBV mode (Leonardo, Siemens). The fused images were then used to assess perfusion defects. All images courtesy of Dr. Kang. |
"The assessment of perfusion defects was done with fused images, and we recorded segments with perfusion defects and counted them," Kang said. "We speculated the cause of the perfusion defects, considering the CT scan."
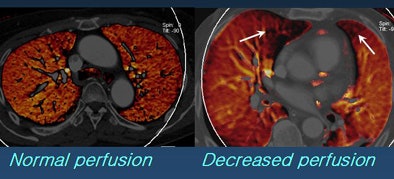 |
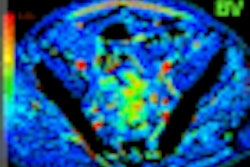
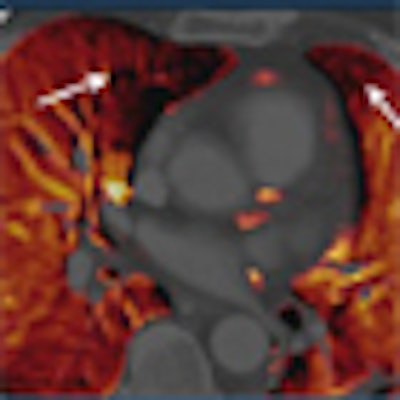
| Image at left shows light red color in both lungs and was interpreted as normal perfusion. However, the fused image at right shows black or dark red colors in the right middle lobe (RML) medial segment and left upper lobe (LUL) lingular segment. These segments were interpreted as decreased perfusion. |
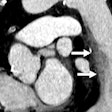
One of the most important artifacts found was beam hardening artifacts due to contrast material in the superior vena cava, Kang said. "This may be the major cause of [perfusion] defects in the [right upper lobe (RUL)] anterior segment (84.6 %), [left upper lobe (LUL)] apical segment (83.3%), and the RUL apical segment (82.1%)," according to Kang.
"Another important cause of perfusion defects is motion artifacts due to cardiac or diaphragmatic motion. We think this may be the main cause of perfusion defects in the [right middle lobe [RML]) medial segment (82.1%), the LUL superior/inferior lingular segments (38.1%/35.7%), and the lung base," she noted.
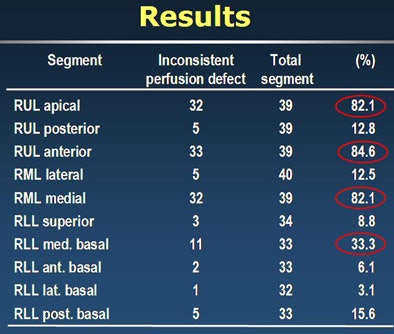 |
| In the right lung (above), perfusion defects were most commonly noted in the right upper lobe (RUL) anterior segment, followed by the RUL apical segment, right middle lobe (RML) medial segment, and right lower lobe (RLL) medial basal segment. In the left lung (below), the most common segment of perfusion defects was the left upper lobe (LUL) apical segment, followed by the LUL superior and inferior lingular segments. |
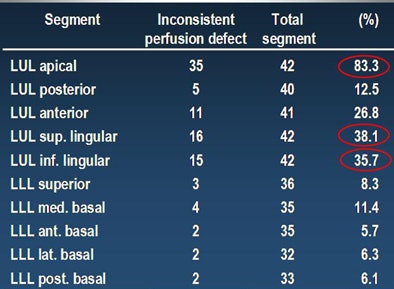 |
The most common locations of perfusion defects not associated with pulmonary embolism were the RUL anterior segment, followed by the LUL apical segment, RUL apical segment, and RML medial segment," Kang said.
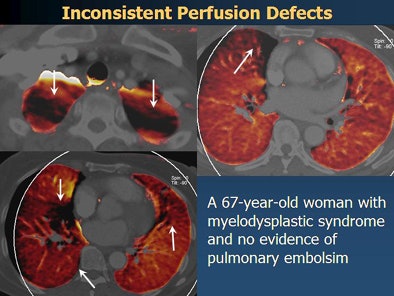 |
| Perfusion defects inconsistent with pulmonary embolism in a 67-year-old woman with myelodysplastic syndrome. There were multifocal perfusion defects in both upper lobe apices, the right middle lobe medial segment, the right lower lobe medial basal segment, and the left upper lobe lingular segment. |
"Knowledge of the inconsistent perfusion defects in nonembolic patients can help us to correctly diagnose pulmonary embolism," Kang said. Of course, optimized protocols are needed to minimize artifacts, and the group has been working to optimize its acquisition methods, she added.
By Eric Barnes
AuntMinnie.com staff writer
December 2, 2008
Related Reading
Multislice CT plus D-dimer testing sufficient for excluding pulmonary embolism, April 21, 2008
Low-dose CT yields equivalent value for PE in pregnancy, November 27, 2007
Researchers recommend alternatives to CT, April 15, 2008
Multicenter trial supports CTPA versus V/Q for PE, December 18, 2007
Pulmonary embolism an uncommon cause of COPD flare, March 6, 2007
Copyright © 2008 AuntMinnie.com