The perfusion exam report [56]:
The perfusion exam report should contain the following information:
1- Indication for the exam: Diagnosis of coronary artery disease (CAD), delineation of extent and severity of CAD, risk stratification, myocardial viability evaluation, or evaluation of acute chest pain syndromes.
2- Clinical history: Patient symptoms, medications (particularly cardioactive medications), and previous diagnostic tests if relevant.
3- Procedure: The type of stress (exercise versus pharmacologic), the adequacy of the stress (heart rate, blood pressure), symptoms during the exam, and reason for termination. Significant ECG findings (ST segment deviations) can also be included.
4- Findings: The quality of the exam should be reviewed- patient motion, subdiaphragmatic/hepatic activity, soft tissue attenuation. Perfusion defects should be described in terms of their location, size (small, medium, and large), type (reversible, fixed, mixed), and severity (mild, moderate, severe). Summed stress, rest, and difference scores may be reported. Abnormal cavity dilatation (with stress or persistent) and abnormal lung activity should be recorded.
5- Impression: State whether the exam is normal or abnormal- and whether there is single or multivessel disease. Probably normal, probably abnormal, and equivocal should be used as infrequently as possible. If the patient is unable to achieve an adequate level of stress or the images are of inadequate quality, the term "non-diagnostic" should be used to describe the study.
Findings indicative of functionally important
coronary artery disease (High risk) [52]:
1- Multiple perfusion defects in more than one coronary artery distribution- reflective of multivessel disease
2- An extensive area of hypoperfusion on stress imaging even if confined to a single vascular distribution (i.e.: proximal LAD pattern). Note that the loaction of the perfusion defect also carries prognostic information [92]. In one study, death or MI occurred in 5.5% of patients with single vessel related ischemia not involving the LAD, 8.4% of patients with single vessel ischemia involving the LAD, and 16.5% of patients with multivessel-related ischemia that also involved the LAD territory [92]. In patients with stable CAD and preserved LV function, the current evidence emphasizes that PCI fails to offer any prognostic benefit over optimal medical therapy, unless a significant amount (generally more than 10%) of the LV myocardium is ischemic [80].
3- Transient ischemic left ventricular cavity dilatation on stress imaging
4- Post stress gated SPECT ejection fraction of less than 40%
5- Increased lung activity on post stress thallium imaging
6- Increased end-diastolic volume or end-systolic volume (greater than 70 mL) as determined by quantitative SPECT
False Positive Stress Perfusion Exam
1. Normal variant
a- Diaphragmatic attenuation
Diaphrgamatic attenuation produces a fixed inferior perfusion defect [50]. It can be exaggerated in obesity and with a full stomach. Diaphragmatic attenuation appears to be more of a problem in men and is estimated to be present in up to 25% of perfusion studies [67]. On ECG gated SPECT, normal function associated with a fixed defect in the inferior wall can be classified as most likely due to soft-tissue attenuation [67]. Although gated imaging helps to assess for wall motion, a problems remains because perfusion defects due to subendocardial infarction that are associated with a normal contraction pattern may be falsely attributed to attenuation artifact [50].
Prone positioning can reduce diaphragmatic attenuation and specificity of the exam by producing an anterior/slight upward shifting of the heart and a lowering of the diaphragm and subdiaphragmatic organs [50,67]. Additionally, prone imaging can substantially reduce the incidence of patient motion due to a more comfortable arm position (usually folded under the patients head) [81]. However, prone imaging may create an anterior or anteroseptal wall perfusion defect and therefore cannot substitute for routine supine imaging (presumably due to sternal or rib attenuation) [50]. Prone imaging may also have value in female patients by correcting breast attenuation [65]. Prone imaging can be performed using 15 seconds per projection with 32 or 64 projections for dual or single-headed cameras respectively [50].
Compared to supine imaging only, the use of combined supine and prone imaging can increased diagnostic certainty, and improve the accuracy and exam specificity without a loss of sensitivity (specificity improved from 65% to 85% and accuracy from 79% to 86%) [60,81,95]. The number of equivocal studies can be reduced from 13% to 4% [95]. Patients with inferior wall defects on supine imaging that are not present on prone imaging have a low risk of subsequent cardiac events (0.7% per year) which is similar to that seen in patients with normal supine exams [50]. To improve efficiency if prone images are done routinely on all patients they should only be acquired only post stress and do not require ECG gating [60]. Acquisition time can also be reduced to 10 minutes [60].
b- Breast shadow or breast implant
Soft tissue attenuation from the breasts may produce artifacts in up to 40% of perfusion studies in women [67]. The breast artifact is most easily appreciated on the rotating tomographic projection or volume rendered images, but can also be seen on planar images. The degree of attenuation varies directly with breast size and tissue density. Classically these defects produce fixed anterior or lateral wall defects, however, any wall can be involved. Remember- body builders with heavily muscled pectoralis muscles have also produced this artifact. Prone imaging may also aid in evaluation of these patients [81]. In one study, compared to supine only imaging, specificity improved from 61% to 94% with combined supine-prone imaging [81]. The largest improvements were noted in patients with the largest breast sizes [81].
c- Pacemakers
Although pacemakers are implanted above and below the diaphragm, it is unusual for a pacemaker to intrude into the chest image and interfere with image interpretation.
2. Mitral
Valve Prolapse
Exercise perfusion defects are uncommon (less than 5%) in patients with mitral valve prolapse, but may be related to exercise induced stimulation of sympathetic activity. This activity produces a tachycardia which will increase the degree of prolapse and consequently stretch the cordae tendinae on the papillary muscles. This stretching may cause ischemia of the papillary muscles and surrounding myocardium. This is only theory, however, and the actual mechanism for these defects is unknown. Since this phenomenon is uncommon, reversible defects in patients with prolapse should still be considered ischemia until proven otherwise. [19]
3.
Valvular Heart Disease
a.Mitral
Regurgitation: Patients
with
regurgitant valvular heart disease may demonstrate myocardial
perfusion abnormalities that are not associated with critical
coronary stenosis at angiography [84].
b. Aortic Stenosis:
Coexisting coronary artery disease is found in about 60% of patients with aortic stenosis [53]. Unfortunately, in patients with aortic stenosis, between 40 to 60% of patients without significant coronary lesions demonstrate either fixed or reversible defects. These perfusion defects are probably related to an increased myocardial oxygen demand combined with a decreased perfusion gradient in the coronary arteries associated with a tight aortic stenosis and left ventricular hypertrophy which produces extravascular compression [40,53]. These perfusion abnormalities may improve postoperatively following the relief of stresses on the pressure loaded ventricle. Pharmacologic stress is preferred in patients with severe aortic stenosis because exercise stress may result in hypotension, syncope, or sudden cardiac death [53].
c. Aortic Regurgitation:
The prevalence of angina pectoris has been reported to vary from 50% to 70% of patients with severe aortic stenosis, however, only half of these patients have coronary lesions at angiography [40]. Patients with aortic regurgitation may demonstrate reversible stress apical defects in the absence of coronary artery disease. With eccentric hypertrophy, the perfusion pressure to the "watershed areas" between the coronary vessels decreases to yield apparent perfusion defects. Perfusion defects elsewhere, should be considered indicative of CAD until proven otherwise.
4. Left
Bundle Branch Block
Patients with left bundle branch block (LBBB) may have a
reversible septal/anteroseptal defect on exercise stress
perfusion imaging that is most pronounced at high heart rates
(14-90% of patients [71]). In LBBB there is delayed septal
activation and relaxation which adversely affects diastolic
coronary flow regionally [54]. With exercise, the myocardium
experiences decreased diastolic perfusion time which yields
decreased radiotracer extraction by the septum relative to the
lateral wall [55]. Technical factors may also contribute to the
finding due to partial volume effect associated with decreased
systolic septal wall thickening [61]. The defect associated with
LBBB should NOT involve the apex- when it does, this favors a
diagnosis of ischemia. In patients with a right ventricular
pacemaker, ventricular conduction originates in the RV and
electrocardiographically mimicks a LBBB (false positive defects
in these patients may also involve the inferior and apical
walls) [87].
Pharmacologic stress with dipyridamole or adenosine which do not increase the heart rate significantly (and therefore should not affect diastolic flow) can be useful in these patients. Despite the greater increase in HR compared to adenosine, regadenoson does not seem to produce septal perfusion defects in patients with LBBB or cardiac pacers [87]. Dobutamine stress, which increases heart rate, can also produce this artifact [41,71].
|
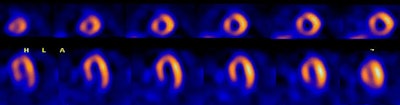
Left bundle branch block: The 50 year old male patient shown below underwent Tc-myoview SPECT imaging following exercise stress. The patients ECG revealed a LBBB. SPECT images showed a perfusion defect involving the septal and anterospetal walls that extended to the apical region of the heart. Rest images were not performed due to a high level of concern for coronary artery disease. The patient's coronary angiogram was normal without evidence of a stenosis. |
|
|
5. Idiopathic Subaortic Stenosis
Increased count density in the region of the thickened septum results in an apparent relative decreased activity in the lateral wall which leads to the impression of a fixed defect in this location. Myocardial perfusion imaging may not be able to reliably distinguish CAD in these patients. Nonetheless, the presence of any type of thallium abnormality in these patients is strongly associated with potentially lethal arrhythmias. (Nucl Med Annual 93, p.214)
6. Hypertensive Myocardial Hypertrophy
Despite concentric myocardial hypertrophy in these patients, there is often increased septal radiotracer activity relative to the lateral wall on both stress and rest images. This increased count density leads to a relative decreased count density in the lateral wall, which may be misinterpreted as a fixed lateral wall abnormality.
7. Cardiac
contusion
8. Infiltrating myocardial
disease
Sarcoid, Amyloid, Neoplasm
9. Cardiomyopathy
a. Dilated Cardiomyopathy
Perfusion defects on rest images can be identified in patients with non-ischemic dilated cardiomyopathies [49] and may be related to partial volume effects associated with thinning of the myocardium with underestimation of the concentration of radioactivity [37]. Another reason for perfusion defects in these patients include areas of myocardial fibrosis [37,49].
b. Hypertrophic Cardiomyopathy:
Ischemia is probably related to an imbalance between oxygen supply in face of the increased myocardial mass, septal perforator arterial compression, abnormalities of intramural arteries, and disturbance of left ventricular relaxation. Reversible perfusion defects are most commonly observed in the most hypertrophied segments of LV - in many cases, there is a mixture of reversible and fixed defects and these abnormalities have been associated with potentially lethal arrhythmias [83]. These patients also demonstrate reversal of the normal lateral-to-septal myocardial count density due to increased septal wall thickness [58]. Image normalization to the hot septum can create apparent fixed lateral wall perfusion defects [58].
Apical hypertrophic cardiomyopathy is characterized by asymmetric myocardial hypertrophy of the apex of the left ventricle [51]. Affected patients generally have a benign clinical course with a low risk for sudden cardiac death [51]. On rest imaging, these patients can demonstrate increased apical tracer uptake, while there is decreased apical activity on stress images even in the absence of coronary artery disease [51]. This is likely related to a disproportionate wall-thickness to vascular supply ration (i.e.: decreased flow reserve) [51].
10.
Bland-White-Garland Syndrome
(Anomalous origin of the left coronary artery from the pulmonary artery)
This anomaly most commonly results in death during early infancy, but survival into adulthood can occur if collateral coronary flow is sufficient. Patients are generally noted to have an anterior wall ischemic perfusion defect. An inferior/posterior perfusion defect may also be seen secondary to a right coronary artery to left coronary artery to pulmonary artery shunt.
11. Coronary spasm
Coronary artery spasm occurs in angiographically normal coronary artery vessels or superimposed on obstructive coronary artery disease (exercise induced spastic angina is more often observed in patients with non-obstructive CAD [97]). Patients with Prinzmetal's angina, cocaine overdoses, and collagen vascular diseases like scleroderma are known to have perfusion defects despite normal coronary arteriograms. Provocative testing with ergonovine during cardiac catheterization may demonstrate latent coronary artery spasm.
12. Myocardial
bridge
In myocardial bridging the coronary artery dips below the epicardium and runs within the myocardium for a short distance [57]. It is a rare angiographic finding (0.5-2.5% of patients at angiography [59]), but the reported incidence at pathologic analysis is higher (15-85%) [57,59]. This discrepancy is likely due to the fact that many affected patients are asymptomatic [59]. Since the LCX and RCA follow the AV groove, myocardial bridging is almost exclusively seen with the LAD- most commonly in the middle segment [59]. Reports have suggested that myocardial bridging may be associated with myocardial ischemia, myocardial infarction, arrhythmias, and sudden death [57]. The characteristic angiographic finding is systolic narrowing of the LAD that disappears during diastole. Exercise stress-induced perfusion defects can be found in 33-88% of patients with myocardial bridging [57]. In general, the greater the degree of vascular compression, the greater the likelihood for perfusion defects [57]. Myocardial bridging that produces a 75% or greater systolic stenosis can demonstrate reversible perfusion defects- even when pharmacologic stress imaging is used [57]. Lesser degrees of narrowing are associated with a lower incidence of perfusion abnormalities [57].
13. Myocarditis
14. Decreased coronary
flow reserve: Syndrome X
Syndrome X is defined as stress-induced anginal pain with a positive stress test for myocardial ischemia, normal findings on coronary angiography, and normal left ventricular function. This is considered a "small vessel disease" syndrome with a reduced coronary vasodilatative reserve of the coronary microcirculation [74]. Its etiology is unclear but may be due to a variable response to exercise and/or pharmacologic stress by the coronary vessels (some dilate more than others) creating an apparent defect. [22] Syndrome X patients are typically found to have higher levels of anxiety [77]. Studies have demonstrated abnormal adrenergic nerve function in the majority of patients with syndrome X by I123 MIBG imaging [74]. Abnormalities on MIBG imaging include severely reduced myocardial uptake and heterogeneous uptake with focal defects [74]. In patients with syndrome X and refractory anginal episodes, spinal cord stimulation has been shown to have beneficial effects- although the beneficial effects do not appear to be related to modulation of adrenergic function [74].
15. Recent
Post-Percutaneous coronary intervention or rotational
atherectomy
Post percutaneous coronary intervention scans should be delayed for at least 2 to 4 weeks after the procedure. Scans performed before this time can reveal apparent fixed defects or are falsely positive for ischemia- these findings may be related to transient persistent myocyte dysfunction or spasm/endothelial damage at the site of the procedure. [31,43]
Transient myocardial perfusion defects can frequently be seen following rotational atherectomy [39]. Etiologies include peripheral vessel obstruction by atheromatous debris, release of vasoactive substances with platelet aggregation, and vessel spasm [39].
16. Papillary
muscles
Insertions of the anterior and posterior papillary muscles may produce focal hot spots typically seen at the 2 (anterolateral) and 7 o'clock positions on short axis images. This finding may be misinterpreted as ischemia of other walls, particularly the inferior.
A corollary to this is the focal hot spots seen on the short axis images in the 7 and 11 o'clock positions in patients with significant right ventricular hypertrophy.
17.
Diaphragmatic Creep
Patients with rapid and deep respiratory excursions following exercise are most likely to demonstrate a gradual upward shift in the position of the heart following exercise which produces an inferior/inferoseptal reversible defect. Waiting 5 -10 minutes prior to imaging generally eliminates this artifact. Cardiac creep is best appreciated on "column mode" sinogram images (or cyclogram) which register "cranocaudal" movement. The tomographic projection images for the column mode sinogram are summed and displayed as if you are facing the patient (coronal images). It is not appreciated on "row mode" sinogram images which register lateral movement. The tomographic projection images for the "row mode" sinogram are summed and displayed as if you were looking down from above the patient (transaxial images). On multiheaded cameras the second detector begins it acquisition at the point just beyond where the first detector completes its movement (between images 32 and 33 for a 64 step acquisition). Diaphragmatic creep will appear as a "jump" at this point in the rotating planar images.
18. Patient Motion
During cardiac SPECT image acquisition the myocardial wall is constantly moving relative to the scanner detectors [66]. Patient motion, respiration, and myocardial contraction all contribute to this motion [66,76]. Respiratory motion is mainly along the axial direction and increases blurring in the anterior and inferior walls [76].
Patient motion is a considerable problem in tomographic imaging
which relies on an accurate center of rotation- for the data to
be appropriately registered, the heart should remain in a
constant position for each projection [62]. Motion will result
in cardiac displacement and misregistration of the projection
data [62]. Motion can introduce image artifacts that can be
erroneously interpreted as perfusion defects [89]. Motion
induced perfusion defects are affected by the type (either
vertical, lateral, or rotational [twisting]) and amount of
motion, motion timing, and the number of camera detectors [27].
The magnitude and timing of the motion determine whether an
artifact will occur, while the direction and pattern of motion
determine the artifacts location [35,62]. Motion artifacts
affect acquisitions with double-headed detectors more than
acquisitions with a single-head detector. This assumption is
related to the fact that a single occurrence of non-returning
motion with a double-headed detector will affect twice as many
projections as with a single-headed detector [27]. However,
acquisitions with a single-headed detector are twice as long as
with double-headed detectors for the same number of collected
counts, and therefore, the chance that the patient will move
during the acquisition is greater [27]. Interestingly, it is
uncommon for motion to mask a perfusion defect (about 1% of
cases) [89].
The heart can develop several characteristic appearances with motion. The "hurricane sign" is seen in the short axis projection- the left ventricle develops the appearance of the National Weather Service symbol for a hurricane and this occurs due to smearing of photon counts in opposing directions around the left ventricle [35]. With motion the heart can also develop a non-rounded, disjointed appearance. Discontinuities of the left ventricular walls, non-anatomic defects, defects at 12 and 6-o'clock, and hot spots are other characteristic patterns of motion artifacts [35,62]. In general, anteroseptal wall defects are more frequently seen with downward motion, anterolateral wall defects are more frequent with upward motion, and inferior wall defects are usually seen with cardiac motion consisting of multiple bounces [35]. Motion correction programs can be applied to diminish artifacts on SPECT images [35].
Factors which affect the final result of motion include [27,28,35]:
1. Amount of movement: Less than 3mm is not usually visually detectable. Movement less than 6.5mm (1 pixel) is detectable, but is not usually clinically significant. Movement of 13mm or greater (2 pixels) frequently produces quantitative abnormalities. Motion correction programs can improve the exam, but studies with greater than 2 pixels of motion should be repeated [27].
2. Type of movement: Vertical movement (up and down) is usually more significant than lateral (horizontal or x-axis) movement. Patient bouncing usually does not produce significant perfusion abnormalities, whereas non-returning motion will create the largest defects [27].
Lateral (horizontal) motion is more
complex and will have a varying effect on the projection images
[27]. For lateral motion, shifting is greatest in the anterior
image (where motion is parallel to the detector) and least in
the lateral image (where motion is perpendicular to the image)
[27]. Horizontal motion characteristically produces perfusion
defects in the septal and lateral walls [89]. Horizontal motion
is usually more difficult to detect in the rotating cinematic
display and sinograms are usually required to detect this type
of motion [89]. In acquisitions without motion, the sinogram
appears as a smooth spiral [89]. Any break or discontinuity in
the septal or lateral margins of the sinogram suggest horizontal
motion [89]. Horizontal motion is also more difficult to correct
with automated motion-corrected software which must detect
discontinuity of the sinogram borders, rather than departure of
the linogram from the horizontal, as is done to correct vertical
motion [89].
Rotational motion- in which the body turns about its axis- can also create artifacts [35].
3. Time and duration of movement: Movement at the beginning or end of a study is less likely to result in image artifacts, whereas movement occurring at mid-aquisition has the worst effect- especially for double-headed detector systems [27]. As a general rule, motion in the projections in which myocardial counts are greater appears to cause larger motion artifacts [27]. Generally, projections in which the camera is closest to the heart will have greater photon counts and will therefore be the most susceptible to motion artifacts [35]. This is because the corresponding projections have higher photon counts, better spatial resolution, and contribute more information to the reconstructed SPECT images [35,62].
Even considerable motion that affects only one or two frames will unlikely result in a perfusion artifact [35]. Motion that corrupts multiple frames is more likely to produce an artifact [35,62].
4. Multiheaded camera: The second detector begins it acquisition at the point just beyond where the first detector completes its movement (between images 32 and 33 for a 64 step acquisition). Therefore, the last projection from detector 1 will appear immediately before the first image from detector 2 when viewing a cine of the projection data [62]. Any movement which occurred during the exam (such as diaphragmatic creep) will appear at this point in the rotating planar images.
Inspection of the projection data in a cine loop format is usually the best way to detect cardiac motion [35, 62]. Sinograms (for horizontal motion) and cyclograms (for verticle motion) are additional tools for motion detection [35]. These images should appear smooth- any discontinuities in the shape of the spirals are reflective of motion [35]. Although significant motion can introduce image artifacts that may be erroneously interpreted as perfusion defects, it is rather uncommon (approximately 1% of cases) for motion to mask a perfusion defect [35].
Motion correction adjusts the projection data to compensate for patient motion [62,89]. The projections are shifted vertically in an attempt to keep the heart in a motion-free trajectory [62]. This shifting can correct for translational motion, but it cannot correct for twisting [62].
Respiration can produce 4-18 mm (average 9mm) of motion in the cranial-caudal direction [66,78]. MPI imaging using high-resolution collimators has an effective resolution of approximately 13 mm (depending on the camera specifications and type of reconstruction) and therefore respiratory motion should have only a minor effect on the quality of MPI images [78]. For 9 mm of motion, the expected impact on the bull's eye map should be less than 10%, however, larger amounts of motion may produce clinically significant changes in the perfusion pattern [66]. Also- respiratory motion can have a greater impact on image quality when high resolution reconstruction algorithms (OSEM-CDR-ACSC) are applied [78].
18. Acquisition
Artifact:
Remember- Perfusion defects occur in the distal tissues subtended by the stenotic artery. Defects involving the proximal septum and/or proximal anterior/anterolateral wall are unlikely to represent ischemia except in patients who have had CABG's. These patients may have high grade occlusions in their native proximal coronary arteries and hemodynamically significant stenoses in their saphenous vein or internal mammary artery grafts.
Abdominal and visceral activity: Intense subdiaphragmatic activity that is adjacent to the heart can produce a variety of artifacts ("intensity artifacts") that alter the observed myocardial activity [38,44]- specifically the ramp-filter artifact and the scatter artifact [86]. There can be an apparent increase in cardiac inferior wall activity due to Compton scatter during acquisition and/or overlap of tracer avid liver or bowel which can mask a true perfusion defect (activity from the bowel "spills over" into the inferior wall) [38,72,86]. The opposite - an apparent perfusion defect in the x-plane of the heart- can also occur as a result of image reconstruction artifact [62,72]. Filtered back projection reconstruction of acquisition images in the presence of intense subdiaphragmatic activity can lead to suppression of adjacent inferior wall counts (a Ramp filter artifact [85]). This occurs during reconstruction when cold/low activity streak artifacts radiated from the intense area of subdiaphragmatic activity into adjacent structures- similar to bladder activity on SPECT bone imaging of the pelvis (backprojection, particularly with the use of a RAMP filter, creates a large band of pixels with negative counts around hot objects during SPECT reconstruction) [44]. These streaks of decreased activity can radiated into the inferior wall and mask tracer uptake [44,46]. This "RAMP filter" artifact can be minimized, but not eliminated, by iterative reconstruction algorithms [62,91]. This type of reconstruction artifact is eliminated by use of wide beam reconstruction [91]. Perfusion defects can also be created due to normalization of the images to "hot" visceral activity (see below).
Normalization and scaling: Interpretation schemes for SPECT
myocardial perfusion imaging usually use a standardized display
in which the stress data set is compared to a resting data set
[47]. Since these data sets are acquired at different times with
different tracer doses, the data sets must be adjusted
(normalized) so that a direct comparison can be performed. Most
commercial systems use series or volume normalization- in this
method, each of these data sets is normalized to the
maximum-intensity pixel (or cluster of pixels) in the entire
data set [47,62]. This permits quantitative comparison of
segmental tracer activity whenever the brightest pixel is
located in the same myocardial region on the resting and stress
images [47]. However, if the area of maximal tracer activity on
a resting study becomes relatively ischemic with stress and is
no longer the brightest region, the comparison of the two exams
loses accuracy [47]. This is because the stress exam will be
normalized "upward" to the brightest pixel in a different
myocardial segment [47]. Re-normalization of regions appearing
to increase with stress to their resting level before image
interpretation can improve detection of multivessel coronary
artery disease [47]. Also- problems occur if the brightest pixel
is located outside of the myocardium- such as the bowel [62]. In
such cases, the heart can appear count-deficient [63]. In frame
normalization, the hottest pixels on the aligned/matched stress
and rest images are identified and set to the maximal intensity
[62]. The next set of matched slices is then evaluated
independently- hence, the scaling problem is not eliminated as
hot spots outside the heart can still result in normalization
artifacts, but this will only affect a specific matched set
[62]. Cardiac normalization is the best method for correctly
normalizing stress and rest data sets [62]. In this method,
software searches only within the myocardium for the maximum
intensity pixel [62]. However, if extracardiac activity lies
within the LV boundry ROI, incorrect normalization can stilll
occur [96].
Translation table: A translation table consists of 4 independent steps- scaling, selecting a color table, selecting the image gamma, and the display media [63]. Scaling consists of two steps- identifying the range of counts to be displayed and determining the step size for each step of the translation table [63]. Color tables can affect image display- defects can appear more pronounced when color scales are used improving sensitivity, but at the expense of specificity [63]. When one encounters a normalization artifact, scaling the affected data set can be performed [62]. For this, the intensity of the images (usually in color) is lowered and then increased until the same maximum color setting is reached on a single area on the stress and rest images [62]. Unfortunately, normalization with this method is somewhat arbitrary and sometimes referred to a "dial-a-lesion".
The image gamma is how the table changes with increasing counts [63]. A logarithmic gamma changes very quickly in the lower count regions and then very slowly in the higher-count areas [63]. This can be useful when reviewing raw tomographic data because it highlights the soft tissue structures around the heart [63]. This type of gamma should never be used for interpreting perfusion images because it will tend to flatten and hide perfusion defects [63]. A sigmoidal gamma tends to increase contrast by emphasizing changes in the mid-count region and flattening changes in the high and low count areas [63]. It can increase sensitivity, but at the expense of specificity [63].
Cardiac position: Cardiac rotation within the chest also affects the apparent tracer distribution. Patients with levorotation have a relative increase in the lateral wall count density, while patients with dextrorotation have a relative increase in the septal wall count density.
The position of the heart in relation to the camera orbit can
also produce artifacts when using 180 degree acquisition orbits.
Although 180 degree SPECT images generally display better image
contrast, reconstruction images are distorted because of
inadequate filtered backprojection [42]. These artifacts of
enhanced contrast and geometric distortion are in part caused by
the non-uniform and depth-dependent spatial resolution and are
further aggravated by the embedded ramp filter in filtered
backprojection reconstruction [42]. For 180 degree orbits and
filtered backprojection reconstruction, an off-center position
of the heart can result in image inhomogeneity with mild
perfusion defects in the anteroseptal and inferolateral walls
(particularly towards the apex) [42]. The inhomogeneity becomes
increasingly more apparent with increasing eccentric cardiac
positioning [42]. The explanation for this finding is that
when a patients heart is not in the center of the orbit, the
gamma heads are at varying distances from the heart during
rotation through the orbit [42]. Segments of the left
ventricular walls closet to the detector are better resolved and
show apparent localized thinning (better resolution), which may
appear as mild perfusion defects [42]. These inhomogeneities are
not observed with 360 degree acquisition orbits [42]. An
interesting consequence of off center cardiac position is that
true perfusion defects will be over-estimated on 180 degree
acquisitions and slightly underestimated by 360 degree
acquisitions [42].
Cardiac translational motion: Cardiac translational motion
between systole and diastole through the fixed imaging plane of
the gamma camera can also lead to an image artifact [90]. In
systole the heart and aortic root recoil downward and medially,
and upward during diastole [90]. As a consequence, this
translational motion may leave the distal inferior imaging plane
with less activity during diastole, thereby creating an
artifactual inferoapical defect that is not present on systolic
images [90].
19. Reversible or Fixed Perfusion Defects in
the Absence of Coronary Artery Disease at Coronary
Angiography:
Reversible perfusion defects have been reported in 20-27% of selected patients with angina and normal findings on coronary angiography [24,64]. In 30-65% of such patients, coronary microvascular dysfunction can be identified as the cause for the perfusion abnormalities [64]. Studies have shown that between 20-40% of diabetic patients, perfusion defects are not found to be associated with obstructive coronary artery disease and these defects are likely related to endothelial dysfunction [79].
Reversible perfusion defects on myocardial perfusion scintigraphy associated with normal coronary angiography may actually represent occult, unrecognized atherosclerotic coronary artery disease which is commonly associated with an abnormal vasodilatory capacity [24]. Importantly, patients with reversible perfusion defects and negative angiography have been shown to have a higher rate of major adverse cardiac events (MACE- in up to 13% of patients over a two year period- a 3.5 times increased risk compared to controls) [73,82]. The prognosis for MACE is significantly worse in patients with a SSS >13 [82]. The presence of chronic renal disease and/or diabetes further increases cardiovascular risk within this subgroup of patients with abnormal MPI, but negative angiographic findings [73,82].
NOTE: The accuracy of myocardial perfusion scintigraphy has previously been evaluated using coronary angiography as the gold standard. Angiography is a silhouette technique that evaluates only the lumenal aspect of the coronary arteries with substantial limitations [24,73]. Work with intracoronary sonography indicates that coronary angiography can underestimate the extent and severity of coronary artery disease- particularly in the early stages of atherosclerosis [24]. Additionally, angiographic underestimation can occur when the reference segment used to quantify the stenosis is also involved by atherosclerotic disease (i.e.: it is not a truly normal segment) [73].
20. Takotsubo cardiomyopathy or Transient
Left Ventricular Apical Ballooning Syndrome:
Transient LV apical ballooning syndrome is a rare form of transient LV dysfunction that mimics an acute coronary syndrome [75]. The disorder is characterized by hypokinesis or dyskinesis of the apical segments and hypercontractility of the basal segments in the absence of atherosclerotic disease [75]. The acute phase of the disorder is felt to be related to metabolic dysfunction at the cellular level [75]. Stress myocardial perfusion imaging during the acute phase will demonstrate a reversible perfusion defect in the apex [75]. Metabolic imaging with FDG has been reported to demonstrate a severe reduction in glucose metabolism in the apical segments (during hyperinsulinemic-euglycemic clamp imaging) [75]. Regional denervation has also been reported in the apex during the acute phase on I123 MIBG imaging [75]. Delayed contrast enhanced MR imaging does not show any evidence of delayed enhancement on post gadolinium imaging [75]. The apical abnormality resolves and normal function and imaging exams can be seen by 3 months following the event [75].
FalseNegative Thallium Exam
Factors associated with the presence of underlying high risk CAD despite normal MPI exam findings include high pre-test probability for CAD (pre-test probability ≥ 66%), typical angina, abnormal TID, abnormal decrease in EF on post stress imaging ( ≥ 5% decrease from rest), or abnormal post stress EF (<43% in men) [93].An important point to remember is that stenoses demonstrated on angiography may not actually be hemodynamically significant or reflect myocardial ischemia, hence, the MPI exam may not be false-negative, but actually a true negative [88]. Development of collateral circulation can provide adequate perfusion to areas of myocardium supplied by a stenotic coronary artery leading to underestimation of the angiographic severity of an epicardial lesion by perfusion imaging [88]. In one study, 18% of coronary stenoses >70% had normal fractional flow reserve [93].
1. Beta
blocker/Calcium channel blocker
Blood pressure medications with antianginal properties (beta-blockers, calcium channel blockers, and nitrates) should be discontinued for at least 48 hours prior to a diagnostic stress test (and more preferably for 4 to 5 half-lives) [70]. Nitroglycerin therapy has been shown to reduce the extent and severity of ischemia assessed by MPI [70]. Submaximal exercise (inability to reach 85% of the maximal predicted heart rate during exercise) results in a lower sensitivity of perfusion imaging for the detection of CAD compared with adequate exercise [31,70]. The negative chronotropic properties of beta-blockers commonly result in submaximal heart rate response to exercise [31]. However, even in patients on beta-blockers that fail to reach 85% of their maximum predicated heart rate, a normal examination may be associated with an overall good prognosis (annual mortality of 0.6% and 0.4% for major ischemic event in one study) [48]. Therefore, when stress imaging is performed to evaluate the effectiveness of anti-ischemic therapy in stable CAD patients, it is reasonable to continue therapy prior to testing [70].
2. "Balanced"
ischemia and left main stenosis
Balanced ischemia is seen in
symmetric three vessel disease- there is relatively balanced
global hypoperfusion which is not recognized without absolute
quantification of regional blood flow [36,93].
|
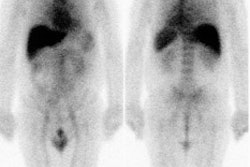
Balanced ischemia: The short axis images below are from an adenosine stress exam- stress images are on top and rest images are below. The exam revealed a prominent left ventricle with fixed perfusion defects in the anterior and posterior walls. No significant reversible perfusion defects were identified. At cardiac catheterization the patient was found to have 3-vessel disease. The case below is an example of "balanced" ischemia. |
|
|
Significant left main stenosis occurs in 5-7% of patients [94]. Medically managed significant left main stenosis is associated with increased cardiac mortality approaching 50% at 3 year followup [94]. Left main stenosis can be difficult to consistently identify on perfusion imaging- particularly if the stenosis is isolated only to this vessel [68]. The left main coronary artery can provide up to 75% of myocardial blood flow, yet defects in both the LAD and circumflex territories are only rarely observed [68]. In one study, moderate to severely abnormal perfusion defects (greater than 10% of the myocardium at stress) were found in only 56% of patients (59% on quantitative analysis) with LM disease [69]. No significant perfusion defects can be identified in 13-19% of patients with LM disease [69,94]. However, TID is observed in a high percentage of patients with LM disease (up to 73%) [69].
In one study, 18% of patients with three-vessel CAD had no perfusion abnormality on MPI and in another study of nonrandomized patients, 36% had evidence of obstructive CAD, despite normal MPI findings [93]. In selected patients with normal MPI, up to one-thrid can have significant CAD [88]. However, some authors suggest that left main or three vessel disease is a less common cause of false-negative MPI in these patients [88]. In their study, single vessel disease was the most common finding (61% of patients- LAD - 40%, RCA - 30%, and LCx - 30%) [88]. In that study, LM disease was the cause of false negative MPI in only 7% of patients and three vessel disease was seen in only 10% [88]. Indirect indicators of three vessel disease include transient LV dilatation and increased lung activity on post stress thallium imaging [36].
3. Insufficient
luminal obstruction
4. Inadequate stress/inadequate vasodilatation
5. Poor technique
Imaging using thallium is delayed
following stress
REFERENCES:
(1) J Nucl Med 1993; Schulman DS, et al. Right ventricular
thallium-201 kinetics in pulmonary hypertension: relation to
right ventricular size and function. 34: 1695-700
(2) New Engl J Med 1993; Zaret BL, Wackers FJ. Nuclear cardiology (1). 329: 775-783
(3) Am J Cardiol 1990; Villanueva FS, et al. Prevalence and correlates of increased lung/heart ratio of thallium-201 during dipyridamole stress imaging for suspected coronary artery disease. 66: 1324-28
(4) Am.J.Cardiol 1990; Lette J, et al. Transient left ventricular cavitary dilation during dipyridamole-thallium imaging as an indicator of severe coronary artery disease. 66:1163-70
(5) J Nucl Med 1994; Iskandrian AS, et al. When is myocardial viability an important clinical issue? 35 (Suppl): 4S-7S
(6) J Nucl Med 1994; Leavitt JI, et al. Demonstration of viable, stunned myocardium with technetium-99m-sestamibi. 35: 1805-07
(7) J Nucl Med 1992; Cuocolo A, et al. Identification of viable myocardium in patients with chronic coronary artery disease: comparison of thallium-201 scintigraphy with reinjection and technetium-99m-methoxyisobutyl isonitrile. 33: 505-511
(8) Circulation 1994; Dilsizian V, et al. Myocardial viability in patients with chronic coronary artery disease. Comparison of 99mTc-sestamibi with thallium reinjection and [18F] fluorodeoxyglucose. 89: 578-87
(9) Radiol Clin North Am 1993; Kiat H, et al. Myocardial perfusion imaging using technetium-99m radiopharmaceuticals. 31: 795-815
(10) J Nucl Med 1994; Altehoefer C, et al. Significance of defect severity in technetium-99m-MIBI SPECT at rest to assess myocardial viability: comparison with fluorine-18-FDG PET. 35: 569-74
(11) J Nucl Med 1995; Maurea S, et al. Enhanced detection of viable myocardium by technetium-99m-MIBI imaging after nitrate administration in chronic coronary artery disease. 36: 1945-52
(12) J Nucl Med 1995; Bisi G, et al. Technetium-99m-sestamibi imaging with nitrate infusion to detect viable hibernating myocardium and predict postrevascularization recovery. 36: 1994-2000
(13) J Nucl Med 1995; Maurea S, et al. Myocardial viability index in chronic coronary artery disease: technetium-99m-methoxy isobutyl isonitrile redistribution. 36: 1953-60
(14) Circulation 1994; 89:578-87
(15) J Nucl Med 1998; Bax JJ, et al. Comparison of Fluorine-18-FDG with rest-redistribution thallium SPECT to delineate viable myocardium and predict functional recovery after revascularization. 39: 1481-1486
(16) J Nucl Med 1995; Ohte N, et al. Clinical significance of
reverse redistribution on 24-hour delayed imaging of exercise
thallium-201 myocardial SPECT: comparison with myocardial
fluorine-18-FDG-PET imaging and left ventricular wall motion.
36: 86-92
(17) J Nucl Med 1993; Liu P, Burns RJ. Easy come, easy go: time to pause and put thallium reverse redistribution in perspective. 34: 1692-94
(18) J Nucl Med 1995; Soufer R, et al. Relationship between reverse redistribution on planar thallium scintigraphy and regional myocardial viability: a correlative PET study.36: 180-87
(19) Nucl Med Annual 1993; Parmett SR, Ongseng. Myocardial perfusion imaging: Pifalls and pearls. Ed. Freeman LM. Raven Press, NY. 195-221
(20) Radiographics 1999; Jadvar H, et al. SPECT and PET in the evaluation of coronary artery disease. 19: 915-926
(21) AJR 2000; Robinson VJB, et al. Causes of transient dilatation of the left ventricle during myocardial perfusion imaging. 174: 1349-1352
(22) Semin Nucl Med 1997; Kataoka T. False-positive myocardial perfusion scintigraphy in syndrome X. 27 (2): 186-189
(23) J Am Coll Cardiol 1996; Mazzanti M, et al. Identification of severe and extensive coronary artery disease by automatic measurement of transient ischemic dilatation of the left ventricle in dual-isotope myocardial perfusion SPECT. 27: 1612-1620
(24) J Nucl Med 2000; Verna E, at al. "False-positive" myocardial perfusion scintigraphy findings in patients with angiographically normal coronary arteries: Insights from intravascular sonography studies. 41: 1935-1940
(25) J Nucl Med 2001; Bax JJ, et al. Relationship between preoperative vaiability and postoperative improvement in LVEF and heart failure symptoms. 42: 79-86
(26) J Nucl Med 2001; Tawakol A, Gewirtz H. Does CABG improve left ventricular ejection fraction in patients with ischemic cardiomyopathy, and does it matter? 42: 87-90 (No abstract available)
(27) J Nucl Med 2001; Matsumoto N, et al. Quantitative assessment of motion artifacts and validation of a new motion correction program for myocardial perfusion SPECT. 42: 687-694
(28) J Nucl Med 1993; Prigent FM, et al. Effect of motion on thallium-201 SPECT studies: a simulation and clinical study. 34:1845-50
(29) J Am Coll Cardiol 1997 Bax JJ, et al. Accuracy of currently available techniques for prediction of functional recovery after revascularization in patients with left ventricular dysfunction due to chronic coronary artery disease: Pooled comparison data. 30: 1451-60
(30) Semin Nucl Med 2000; Bas JJ, et al. 18-Fluorodeoxyglucose imaging with positron emission tomography and single photon emission computed tomography: Cardiac applications. 30: 281-298
(31) Semin Nucl Med 1995; Newhouse HK, Wexler JP. Myocardial perfusion imaging for evaluating interventions in coronary artery disease. 25: 15-27
(32) J Nucl Med 2001; Tanaka R, Nakamura T. Time course evaluation of myocardial perfusion after reperfusion therapy by 99mTc-tetrofosmin SPECT in patients with acute myocardial infarction. 42: 1351-1358
(33) J Nucl Med 2001; Harel F, et al. Clinical impact of combination of scatter, attenuation correction, and depth-dependent resolution recovery for 201Tl studies. 42: 1451-56
(34) Radiology 2001; Oshinski JN, et al. Quantitative prediction of improvement in cardiac function after revascularization with MR imaging and modeling: Initial results. 221: 515-522
(35) J Nucl Cardiol 2001; Fitzgerald J, Danias PG. Effect of motion on cardiac SPECT imaging: recognition and motion correction. 8: 701-706
(36) J Nucl Med 2002; Yamagishi H, et al. Incremental value of left ventriuclar ejection fraction for detection of multivessel coronary artery disease in exercise 201Tl gated myocardial perfusion imaging. 43: 131-139
(37) J Nucl Med 2002; Hassan N, et al. 201Tl SPECT abnormalities, documented at rest in dilated cardiomyopathy, are related to a lower than normal myocardial thickness but not to an excess in myocardial wall stress. 43: 451-457
(38) J Nucl Cardiol 2002; Samady H, et al. Pharmacologic stress perfusion imaging with adenosine: Role of simultaneous low-level treadmill exercise. 9: 188-196
(39) J Nucl Cardiol 2002; Koch KC, et al. Mechanisms of myocardial hypoperfusion during rotational atherectomy of de novo artery lesions and stenosed coronary stents: insights from serial myocardial scintigraphy. 9: 304-311
(40) J Nucl Cardiol 2002; L'Abbate A, et al. Myocardial perfusion and coronary microcirculation: from pathophysiology to clinical application. 9: 328-337
(41) J Nucl Med 1994; Tighe DA, et al. False-positive reversible perfusion defect during dobutamine-thallium imaging in left bundle branch block. 35:1989-91
(42) J Nucl Med 2002; Liu YH, et al. Differential effect of 180 degrees and 360 degrees acquisition orbits on the accuracy of SPECT imaging: quantitative evaluation in phantoms. 43: 1115-1124
(43) J Nucl Cardiol 2002; Angiolillo DJ, Giordano A. Role of myocardial perfusion imaging after coronary revascularization in symptom-free patients: are low-risk patients really low? 9: 550-553
(44) J Nucl Cardiol 2002 Funahashi M, et al. Application of pixel truncation to reduce intensity artifacts in myocardial SPECT imaging with Tc-99m tetrofosmin. 9: 622-31
(45) J Nucl Cardiol 2003; De Lorenzo A, et al. Use of atropine in patients with submaximal heart rate during exercise myocardial perfusion SPECT. 10: 51-55
(46) J Nucl Cardiol 2003; Boz A, et al. The effects of solid food in prevention of intestinal activity in tc-99m tetrofosmin myocardial perfusion scintigraphy. 10: 161-167
(47) J Nucl Cardiol 2003; Williams KA, et al. Correct spatial normalization of myocardial perfusion SPECT improves detection of multivessel coronary artery disease. 10: 353-360
(48) J Nucl Cardiol 2003; Marie PY, et al. Residual exercise SPECT ischemia on treatment is a main determinant of outcome in patients with coronary artery disease treated medically at long-term with beta-blockers. 10: 361-368
(49) J Nucl Cardiol 2003; Wu YW, et al. Tl-201 myocardial SPECT in differentiation of ischemic from non-ischemic dilated cardiomyopathy in patients with left ventricular dysfunction. 10: 369-374
(50) J Nucl Med 2003; Hayes SW, et al. Prognostic implications of combined prone and supine acquisitions in patients with equivocol or abnormal supine myocardial perfusion SPECT. 44: 1633-1640
(51) J Nucl Cardiol 2003; Ward RP, et al. Resting "solar polar" map pattern and reduced apical flow reserve: characteristics of apical hypertrophic cardiomyopathy on SPECT myocardial perfusion imaging. 10: 506-512
(52) J Nucl Cardiol 2003; Beller GA. Clinical value of myocardial perfusion imaging in coronary artery disease. 10: 529-542
(53) J Nucl Cardiol 2004; Patsilinakos SP, et al. Adenosine stress myocardial perfusion tomographic imaging in patients with significant aortic stenosis. 11: 20-25
(54) J Nucl Cardiol 2004; Kasai T, et al. Decreased septal thickening in patients with left bundle branch block. 11: 32-37
(55) J Nucl Cardiol 2004; Hansen CL. The conundrum of left bundle branch block. 11: 90-92
(56) J Nucl Cardiol 2003; Hendel RC, et al. American society of nuclear cardiology consensus statement: reporting of radionuclide myocardial perfusion imaging studies. 10: 705-708
(57) J Nucl Cardiol 2005; Vallejo E, et al. Myocardial perfusion SPECT imaging in patients with myocardial bridging. 12: 318-323
(58) J Nucl Cardiol 2005; Ghai A, et al. Significant left ventricular hypertrophy on SPECT imaging in a patient with hypertrophic cardiolyopathy: a comparative image with cardiac magnetic resonance. 12: 605-606
(59) Radiographics 2006; Kim SY, et al. Coronary artery anomalies: classification and ECG-gated multi-detector row CT findings with angiographic correlation. 26: 317-334
(60) J Nucl Cardiol 2006; Berman DS, et al. Diagnostic accuracy of gated Tc-99m sestamibi stress myocardial perfusion SPECT with combined supine and prone acquisitions to detect coronary artery disease in obese and nonobese patients. 13: 191-201
(61) J Nucl Cardiol 2006; Zupan K, et al. Thallium-201 myocardial SPECT in left bundle branch block: diagnosis of myocardial ischemia with a disease-specific reference database. 13: 521-530
(62) J Nucl Cardiol 2006; Ibrahim DY, et al. Optimal SPECT processing and display: making bad studies look good to get the right answer. 13: 855-866
(63) J Nucl Cardiol 2006; Hansen CL. The role of the translation table in cardiac image display. 13: 571-575
(64) J Nucl Med 2007; Graf S, et al. Typical chest pain and normal coronary angigram: cardiac risk factor analysis versus PET for detection of microvascular disease. 48: 175-181
(65) J Nucl Cardiol 2007; Slomka PJ, et al. Combined quantitative supine-prone myocardial perfusion SPECT improves detection of coronary artery disease and normalcy rates in women. 14: 44-52
(66) J Nucl Med 2007; Kovalski G, et al. Correction of heart motion due to respiration in clinical myocardial perfusion SPECT scans using respiratory gating. 48: 630-636
(67) J Nucl Cardiol 2007; Singh B, et al. Attenuation artifact, attenuation correction, and the future of myocardial perfusion SPECT. 14: 153-164
(68) J Nucl Cardiol 2007; Travin MI. Is it possible for myocardial perfusion imaging to avoid missing any patients with high-risk coronary disease? 14: 492-496
(69) J Nucl Cardiol 2007; Berman DS, et al. Underestimation of extent of ischemia by gated SPECT myocardial perfusion imaging in patients with left main coronary artery disease. 14: 521-528
(70) J Nucl Cardiol 2007; Baghdasarian SB, et al. Patient preparation for nuclear imaging: when should anti-ischemic medications be withheld? 14: 775-781
(71) J Nucl Med 1997; Caner B, et al. Dobutamine thallium-201 myocardial SPECT in patients with left bundle branch block and normal coronary arteries. 38: 424-427
(72) J Nucl Cardiol 2008; Thompson RC. The problem of radiotracer abdominal activity in myocardial perfusion imaging studies. 15: 159-161
(73) J Nucl Cardiol 2008; Alqaisi F, et al. Prognostic predictors and outcomes in patients with abnormal myocardial perfusion imaging and angiographically insignficant coronary artery disease. 15: 754-761
(74) J Nucl Cardiol 2008; Spinelli A, et al. Effect of spinal cord stimulation on cardiac adrenergic nerve function in patients with cardiac syndrome X. 15: 804-810
(75) J Nucl Cardiol 2008; Feola M, et al. Reversible impairment of coronary flow reserve in takotsubo cardiomyopathy: a myocardial PET study. 15: 811-817
(76) J Nucl Cardiol 2009; Kovalski G, et al. Dual "motion-frozen heart" combining respiration and contraction compensation in clinical myocardial perfusion SPECT imaging. 16: 396-404
(77) J Nucl Cardiol 2009; Vermeltfoort IAC, et al. Association between anxiety disorder and the extent of iscehmia observed in cardiac syndrome X. 16: 405-410
(78) J Nucl Cardiol 2009; Kovalski G, et al. Correction for respiration artifacts in myocardial perfusion SPECT is more effective when reconstructions supporting collimator response compensation are applied. 16: 949-955
(79) J Nucl Med 2009; Djaberi R, et al. Endothelial dysfunction in diabetic patients with abnormal myocardial perfusion in the absence of epicardial obstructive coronary artery disease. 50: 1980-1986
(80) J Nucl Cardiol 2010; Gaemperli O, Kaufmann PA. Multimodality cardiac imaging. 17: 4-7
(81) J Nucl Cardiol 2010; Germano G, et al. Supine acceptance of a conventional imaging position may make you less prone to success. 17: 16-18
(82) J Nucl Cardiol 2010; Adamu U, et al. Stress/rest myocardial perfusion scintigraphy in patients without significant coronary artery disease. 17: 38-44
(83) J Nucl Cardiol 2011; Shirani J, Dilsizian V. Nuclear cardiac imaging in hypertrophic cardiomyopathy. 18: 123-134
(84) J Nucl Cardiol 2011; Sidhu M, et al. Myocardial perfusion
imaging analysis in patients with regurgitate valvular heart
disease. 18: 309-313
(85) J Nucl Cardiol 2012; DePuey EG. Advances in SPECT camera
software and hardware: currently available and new on the
horizon. 19: 551-552
(86) J Nucl Cardiol 2013; Case JA, Bateman TM. Taking the
perfect nuclear image: quality control, acquisition, and
processing techniques for cardiac SPECT, PET, and hybrid
imaging. 20: 891-907
(87) J Nucl Cardiol 2013; Thomas GS, et al. Is regadenoson an
appropriate stressor for MPI in patient with left bundle branch
block or pacemakers? 20: 1076-1085
(88) J Nucl Cardiol 2014; Yokota S, et al. Prevalence,
location, and extent of significant coronary artery disease in
patients with normal myocardial perfusion imaging. 21: 284-290
(89) J Nucl Cardiol 2014; Agarwal V, DePuey EG. Myocardial
perfusion SPECT horizontal motion artifact. 21: 1260-1265
(90) J Nucl Med 2015; Kitkungvan D, et al. Clinical utility of
enhanced relative activity recovery on systolic myocardial
perfusion SPECT: lessons from PET. 56: 1882-1888
(91) J Nucl Cardiol 2016; Shah A, et al. Intense moving
intestinal activity as a source of artifact on myocardial
perfusion SPECT study. 23: 161-167
(92) J Nucl Cardiol 2016; Hage FG, et al. Review of
cardiovascular imaging in the Journal of Nuclear Cardiology in
2015- Part 2 of 2: myocardial perfusion imaging. 23: 493-498
(93) J Nucl Cardiol 2016; Nakanishi R, et al. Predictors of
high-risk coronary artery disease in subjects with normal SPECT
myocardial perfusion imaging. 23: 530-541
(94) J Nucl Cardiol 2016; Sareen N, Ananthasubramaniam K. Left
main coronary artery disease: a review of the spectrum of
noninvasive diagnostic modalities. 23: 1411-1429
(95) J Nucl Cardiol 2016; Taasan V, et al. Comparative accuracy
of supine-only and combined supine-prone myocardial perfusion
imaging in men. 23: 1470-1476
(96) J Nucl Cardiol 2017; Malek H, et al. Artifacts in
quantitative analysis of myocardial perfusion SPECT, using
Cedars-Sinai QPS software. 24: 534-542
(97) J Nucl Cardiol 2017; Songy B. Detection of non-obstructive coronary artery disease: is post-stress diastolic dysfunction assessed by myocardial perfusion imaging a useful tool? 24: 1551-1554