A slate of new x-ray systems, including a digital radiography system, a dual-purpose radiography and fluoroscopy system, and a mobile C-arm, are among the highlights in the virtual booth of Siemens Healthineers at this week's RSNA 2020 virtual meeting.
Radiography
Multix Impact C is a new ceiling-mounted system in the company's Multix family of digital radiography (DR) systems. Siemens is also launching Multix Impact VA20, a new version of its floor-mounted DR room.
Both Impact C and Impact VA 20 include MyExam Companion, the company's software that guides radiologic technologists and radiographers through exam setup and image acquisition. The software is designed to provide a user interface that can help x-ray personnel identify the optimal acquisition and reconstruction parameters, based on the patient's gender and age, as well as other patient-specific information.
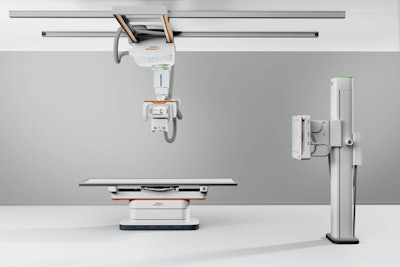 Multix Impact C is a new ceiling-mounted system in the company's Multix family of DR systems. All Images courtesy of Siemens.
Multix Impact C is a new ceiling-mounted system in the company's Multix family of DR systems. All Images courtesy of Siemens.MyExam Companion includes Smart Visual Ortho, a feature that helps staff set the exam's field-of-view and adjust exposure parameters on a touchscreen using images from a live camera mounted in the x-ray room. This can make long-leg and full-spine exams easier to set up and conduct, according to the company.
Another feature uses artificial intelligence-based detection and collimation of body parts to speed up workflow for spine and leg exams by analyzing the contours of a patient and adjusting collimator blades, all in less than half a second, according to the company.
Both Multix Impact C and Multix Impact VA 20 are pending 510(k) clearance, according to Siemens.
Meanwhile, Luminos Lotus Max is a combined fluoroscopy and radiography system designed to enable users to perform both dynamic and static x-ray exams. In launching the system, Siemens noted that the number of fluoroscopy studies being performed is declining, so users want a system that is also suited for radiography -- users can switch quickly between the modes, according to the company.
The remote-controlled Lotus Max can be operated from a control room, and it can also be operated from controls located at the system's table. It's capable of performing specialized orthopedic studies like long-leg or full-spine exams, while also performing basic interventional radiology procedures.
Options available for Lotus Max include a wireless foot switch that can potentially reduce operator radiation dose, as well as a package of software for reducing radiation dose to patients, improving workflow, and improving image quality. What's more, a multicolor MoodLight feature creates a more hospitable environment for patients and staff.
Interventional
In the realm of mobile C-arms, Cios Flow is a new offering from Siemens that's designed to increase the efficiency of routine surgical interventional procedures, according to the company. The system is designed to be used in a variety of clinical settings, including orthopedics, trauma surgery, spinal surgery, vascular surgery, and pain therapy.
Siemens is highlighting the low weight, maneuverability, and touch-gesture interface of the C-arm. It also includes a function called SpotAdapt that helps users visualize areas of challenging anatomy by allowing them to tap an area on a preview image of the user interface; the technique automatically optimizes parameters such as brightness and contrast.
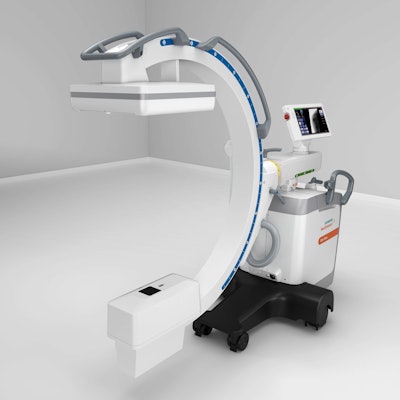 Cios Flow is a mobile C-arm designed to increase the efficiency of routine surgical interventional procedures.
Cios Flow is a mobile C-arm designed to increase the efficiency of routine surgical interventional procedures.Siemens has also built new security functions into Cios Flow to reduce the risk of cyberattacks. Additionally, it has user access and management features to block unauthorized access to sensitive patient data, and patient data can be encrypted using BitLocker software. Applications on Cios Flow can also be "whitelisted" to ensure that only trusted sources can access them.
Cios Flow and the SpotAdapt feature are pending 510(k) clearance and are not yet commercially available in the U.S.
Siemens is also showing new capabilities for its Corindus product line, which it accessed with its acquisition of Corindus Vascular Robotics in 2019. The division is launching a new technology called TechnIQ, which enables the CorPath GRX system to perform movements that mimic gestures made by operators during interventional procedures. These include "rotate on retract," "wiggle," "spin," "dotter," and "constant speed." The movements can be performed for applications like guidewire navigation and lesion crossing and anatomy measurement.
MRI
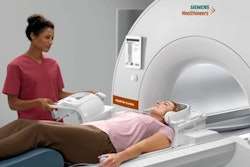
In the MRI section of the Siemens booth, the company is highlighting the work-in-progress Magnetom Free.Max, a midfield scanner at 0.55-tesla with a wide 80-cm bore. The scanner does not have 510(k) clearance from the U.S. Food and Drug Administration (FDA).
Free.Max is designed to be a small, lightweight scanner that can be easily installed at multiple locations, bringing MRI to clinical settings where it previously hasn't been used. The system also needs only 0.7 L of helium at installation, and it never requires helium refills, meaning it can be sited without the installation of a quench pipe.
The scanner employs the same user interface as other MRI systems in the Magnetom product line, and it also employs Siemens' myExam Companion technology for assisting radiographers with setting up and performing exams using standardized protocols.
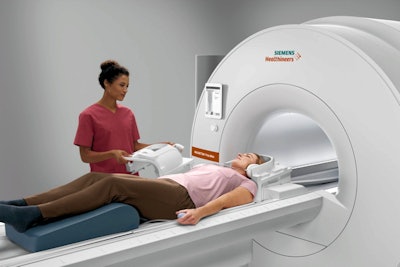 Magnetom Free.Max is a 0.55-tesla MRI scanner being shown as a work-in-progress.
Magnetom Free.Max is a 0.55-tesla MRI scanner being shown as a work-in-progress.The company is discussing two new applications based on artificial intelligence (AI) and designed to provide assistance in the interpretation of MR images, one for the brain and the other for the prostate.
AI-Rad Companion Brain MR for Morphometry Analysis automatically measures volumetric changes in the brain through brain volumetry, which involves measuring the volume of gray matter, white matter, and cerebrospinal fluid. Previously, the technique was performed manually or semiautomatically. Lower volume can indicate the presence of Parkinson's or Alzheimer's disease.
The other application, AI-Rad Companion Prostate MR for Biopsy Support, segments the prostate on MR images and marks the organ's outer contour in seconds, much faster than manual segmentation. Radiologists can then mark suspicious regions and send annotated MR images to urologists for fusion with ultrasound images during biopsy procedures. Both applications received FDA clearance earlier this year.
Meanwhile, Deep Resolve is an AI-based image reconstruction technique designed to generate high-resolution images from low-resolution data input. It enables faster MRI scans, which can improve patient comfort and boost departmental workflow, with no sacrifice in resolution, according to the company.
Siemens is developing Deep Resolve for its 1.5-tesla and 3-tesla MRI scanners
CT
New applications are the focus of the CT area of Siemens' booth at RSNA 2020. The company is demonstrating its myExam Companion technology for guided exam setup with CT studies. The technique can help radiologic technologists and radiographers in the setup and performance of CT exams, and it can alert them to complications such as whether a patient has a metal implant.
The company is also demonstrating its Somatom On.site portable CT scanner, which was first launched at the RSNA 2019 conference. In developing the system, Siemens mounted a smaller CT scanner gantry on a portable motorized trolley that can be moved to a patient's bedside. The 32-slice scanner has a 0.7-mm detector width, providing 2.4 cm of coverage per gantry rotation.
In addition, On.site has an integrated camera that supports real-time viewing on the built-in touch display, and the scanner's telescopic gantry design allows the radiation source to move away from the patient during scanning. Images acquired with On.site can be sent to PACS wirelessly. On.site was cleared by the FDA earlier this year.
Finally, Siemens is demonstrating CT for the assessment of structural heart disease. The company believes that its dual-source CT technology has unique advantages for heart imaging.
Molecular imaging
In the molecular imaging section of its booth, Siemens is highlighting Cerianna (fluoroestradiol F-18), a radiopharmaceutical for PET imaging of patients with metastatic breast cancer. The radiopharmaceutical was approved by the FDA earlier this year and is the result of a collaboration between Siemens and Zionexa USA.
Cerianna is the first FDA-approved F-18 radiopharmaceutical specifically indicated for use in PET detection of estrogen receptor-positive lesions as an adjunct to biopsy in patients with recurrent or metastatic breast cancer, according to Siemens and its PETNet Solutions subsidiary. Cerianna will be commercially available in late 2020 or early 2021.
In other molecular imaging news, Siemens is also highlighting OncoFreeze AI, a data reconstruction tool in the company's AIDAN family that's designed to reduce the impact of respiratory motion on PET/CT oncology images of the chest or abdomen.
With OncoFreeze AI reconstruction, 100% of acquired PET counts are used for image reconstruction, according to Siemens. This means that no additional scanning time is required for a motion-frozen image.
For SPECT, Siemens is discussing its xSPECT Quant technique, which provides quantitative SPECT/CT images. The technique is available for use with a variety of radiopharmaceuticals.
Ultrasound
New software releases are being highlighted for the company's lineup of Acuson ultrasound scanners, which include the Sequoia, Redwood, and Juniper systems. A new technology being highlighted is slow flow, a Doppler technique that allows users to easily visualize more vessels that are smaller, helping users see deeper into tissue, according to the company.
Siemens is also discussing new transducers in a version 1.1 software release for the Sequoia flagship scanner, as well as a low-frequency linear transducer and a 4D endocavity probe. The package has been cleared by the FDA and is in commercial release.
For the value-tier Juniper scanner, Siemens has issued a 1.5 software release that has shared-service capabilities for cardiac imaging, including the company's vascular burden index (VBI) technique for strain measurement in the heart.
Imaging informatics and AI
As a work-in-progress, Siemens is discussing Syngo Carbon, a new software environment for enterprise-wide interpretation and reporting of medical images. Syngo Carbon integrates digital data from different hospital departments and presents it within a unified environment, simplifying workflow and making it easier for clinicians to work together.
Unlike traditional PACS, Syngo Carbon works with all types of medical image data, not just DICOM files, and includes files from pathology, endoscopy, and cardiology. Syngo Carbon users can continue to use their existing technologies and data, according to the company. Syngo Carbon also will enable users to view PACS and advanced visualization data with the same workstation and interface.
Siemens is about to begin customer testing of Syngo Carbon, with commercial shipments sometime in mid-2021.
In the artificial intelligence space, Siemens is highlighting the AI-Rad Companion family of applications, which are being displayed in various areas in the company's booth.
Siemens has taken a multiorgan approach with AI-Rad Companion, such as the AI-Rad Companion Brain MR for Morphometry Analysis and the AI-Rad Companion Prostate MR for Biopsy Support applications, found in the MRI section of the company's booth.
The newest application in the family is AI-Rad Companion Chest X-Ray, which can detect suspicious findings on chest x-ray exams acquired with x-ray systems from multiple vendors. The application analyzes thoracic radiography images for findings like pneumothorax and alerts physicians so that these patients can be seen more quickly. The application has received the CE Mark; it is being shown as a work-in-progress.
Siemens is demonstrating a dashboard designed to help staff analyze the quality of x-ray images by providing data such as rejected image analysis and radiation exposure index.
Finally, Siemens is highlighting the recent FDA clearance of its AI-Rad Companion Organs RT application, designed for guiding radiation therapy treatment. The application automatically contours healthy organs that could be at risk during radiation therapy treatments, based on deep-learning algorithms.
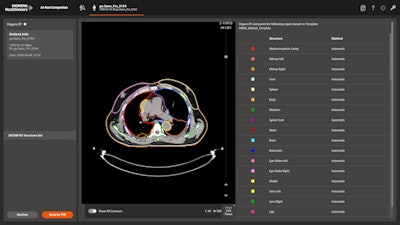 AI-Rad Companion Organs RT is an AI-based application for guiding radiation therapy treatment. Image courtesy of Siemens.
AI-Rad Companion Organs RT is an AI-based application for guiding radiation therapy treatment. Image courtesy of Siemens.The application is designed to replace manual organ contouring, which can be a time-consuming task for radiation oncologists. The technique works across multiple organs, including the head and neck, thorax, abdomen, and pelvic regions.
Women's imaging
In the women's imaging section of its booth, Siemens is pointing to recent enhancements to its line of mammography systems, including Mammomat Inspiration and Mammomat Fusion. A major point of emphasis for the company is Breast Health 360°, the company's breast care concept that follows every step of a woman's journey through the breast health cycle, from diagnosis to recovery.