CHICAGO - A new portable head CT scanner on wheels, artificial intelligence (AI) tools for MRI, and a premium shared-services ultrasound scanner are among the RSNA 2019 highlights for Siemens Healthineers this week.
CT
Somatom X.cite is a new CT scanner that Siemens launched a few weeks prior to RSNA 2019. The scanner will be the company's flagship single-source CT system.
Somatom X.cite is the first to use what Siemens calls myExam Companion, an intelligent user interface concept that identifies the optimal acquisition and reconstruction parameters for patients using information such as their gender and age.
MyExam Companion is based on AI analysis of almost 200,000 scan parameters and the work of clinical experts who identified scan strategies and clinical decision trees. It includes more than 20 clinical decision trees that can be defined by users and are displayed at either the scanner operator console or on mobile detachable tablets that can be used to control the scanner.
X.cite features an 82-cm bore for improved patient comfort, and it also uses the company's Vectron x-ray tube, which previously was only available on the dual-source Somatom Force. The scanner also includes the company's Fast 3D overhead camera to help position patients.
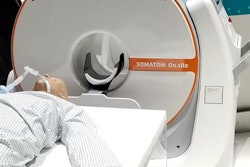
Siemens is also using RSNA 2019 to launch Somatom On.site, a portable head scanner for use in the intensive care unit (ICU). Previously, ICU patients had to be transported to the radiology department for scans -- a challenging prospect for both patients and hospital staff.
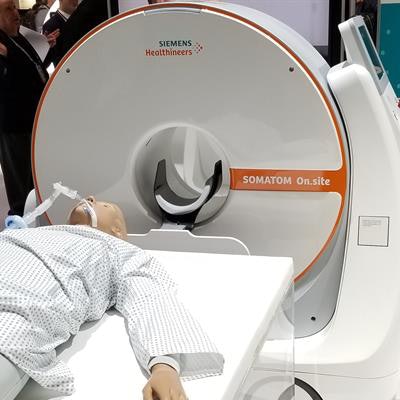
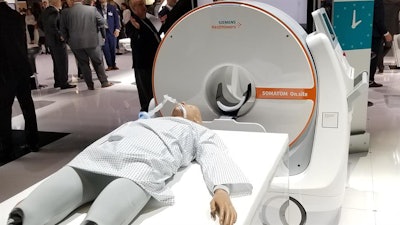 Somatom On.site is a new portable head CT scanner that Siemens is launching at RSNA 2019.
Somatom On.site is a new portable head CT scanner that Siemens is launching at RSNA 2019.On.site enables the CT scanner to be moved to the patient's bedside. The scanner sites on a portable motorized trolley that can be easily moved with a handle, and images can be sent to PACS wirelessly. The 32-slice scanner has a 0.7-mm detector width, providing 2.4 cm of coverage per gantry rotation.
On.site is pending 510(k) clearance from the U.S. Food and Drug Administration (FDA), and shipments are expected to begin in the spring of 2020. MyExam Companion is also available on the system.
MRI
In MRI, Siemens is discussing new AI-based software for its AI-Rad Companion line.
AI-Rad Companion Brain MR is designed for morphometry analysis and automatically segments MR brain images, measures brain volume, and marks volume deviations in tables for diagnosis and treatment.
Through its AI algorithms, AI-Rad Companion Brain MR identifies approximately 30 brain segments on MR images and compares the results with data in a reference database for brain morphometry from the Alzheimer's Disease Neuroimaging Initiative (ADNI).
In cases of suspected dementia, Alzheimer's disease, and Parkinson's disease, the brain volumetry function quantifies changes in the volume of gray and white matter, as well as cerebrospinal fluid, which serve as biomarkers for the conditions.
Abnormal results are automatically tagged in a patient's report to give radiologists objective, quantitative data relevant to the diagnosis and subsequent therapy management.
Similarly, Siemens' AI-Rad Companion Prostate MR automatically segments MR images of the prostate so radiologists can mark lesions and the organ's outer contour for more precise procedures and enhanced patient outcomes.
Annotated MR images are sent to the urologist for fusion with ultrasound images during the biopsy, helping to detect significant prostate carcinomas.
Both AI-Rad Companion Brain MR and AI-Rad Companion Prostate MR are pending 510(k) clearance and are designed for use on any vendor's MRI scanner.
Siemens is also touting its syngo Virtual Cockpit software for MRI, which allows radiologists and other specialists to connect remotely to MRI, CT, PET/CT, and PET/MRI scanners to help image patients who require more sophisticated or complex exams.
On the hardware side, the company is showing Magnetom Lumina, a 3-tesla MRI scanner that received FDA clearance earlier this year.
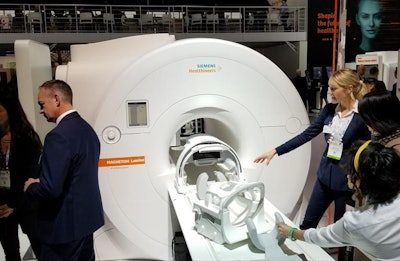 Siemens is showing the Magnetom Lumina MRI scanner this week at RSNA 2019.
Siemens is showing the Magnetom Lumina MRI scanner this week at RSNA 2019.Ultrasound
Acuson Redwood is a new shared-services premium ultrasound scanner that Siemens launched in October. The system features advanced radiology applications such as contrast-enhanced ultrasound (CEUS) and shear-wave elastography (SWE), as well as cardiology capabilities.
Redwood is based on Siemens' coherent image formation (CIF) technology, designed to maintain B-mode image quality even in complex scanning modes. In addition, it employs the company's UltraArt universal image processing technology.
Redwood's cardiac-oriented features include syngo Velocity Vector Imaging (VVI), stress echocardiography with a wall motion scoring analysis package, and a left ventricular opacification mode to support cardiac contrast imaging.
Redwood has FDA clearance and will begin shipping in one to two months. It shares the same probe family as the Acuson Sequoia scanner.
Interventional
In the interventional section of its booth, Siemens is highlighting the Artis icono family of angiography systems, which were launched at the 2019 edition of the European Congress of Radiology (ECR).
Artis icono is intended to support both radiology and cardiology interventional procedures using a novel detector positioning design. The family features a new detector positioning system called Lateral Plane Switch that lets users switch from the optimal radiology configuration to the best cardiology setup, and vice versa, in just seconds.
Siemens also redesigned its angiography imaging chain for the Artis icono systems. Called Optiq, the imaging chain includes redesigned image processing, ranging from acquisition to postprocessing to on-screen display.
All icono units use Siemens' newest 16-bit high dynamic range (HDR) digital detectors and come in two detector sizes: a 30 x 40-cm detector for radiology and neuro work and a new 21 x 21-cm detector for cardiology procedures.
At RSNA 2019, Siemens is showing an Artis icono system integrated with an endovascular robot from Corindus, a company that Siemens acquired in October. It is also demonstrating the system with a Mentice simulator for training physicians to perform interventional procedures.
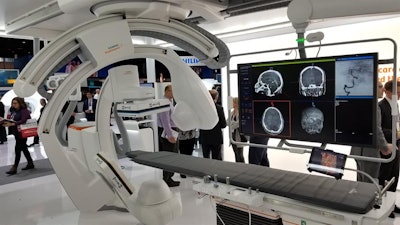 Siemens is showing its Artis icono angiography system integrated with a Corindus endovascular robot.
Siemens is showing its Artis icono angiography system integrated with a Corindus endovascular robot.Artis icono has received 510(k) clearance and is expected to begin shipping in early 2020.
X-ray
In the x-ray section of the company's booth, Siemens is highlighting new optimization modes for musculoskeletal imaging on its Multitom Rax system, including a Smart Orbital Lateral mode.
For Multix Impact, the company is demonstrating a new full-size 17 x 17-inch detector, as well as a new dual-detector configuration, with one wireless detector and one in a wall stand. The two-detector version of Multix Impact is pending 510(k) clearance and will begin shipping in 2020.
Finally, Siemens is showing the ability to control the Multix Impact system not only at the tube head but also using a tablet computer.
Human resources
Siemens is also debuting a cloud-based platform designed to help healthcare facilities better address staffing shortages and maintain workforce and productivity levels.
CrewPlace is a digital portal through which offsite or in-house radiologists and technologists can be summoned and assigned to areas where their expertise is needed.
With the portal, facilities can match a technologist's workday and time preferences with staffing requirements. CrewPlace can also dispatch a technologist to a location within the organization where a particular procedure is being performed and his or her skillset is needed.
The platform is available free of charge to customers purchasing new advanced therapies fixed systems, including the Artis icono angiography system, and existing systems covered by the company's service contract with advanced case support.
It is currently in place at 10 facilities, primarily in the southern half of the U.S.