Monitor distributor Double Black Imaging (DBI) and its Image Systems division is previewing their new 12-megapixal (MP) LED backlit display at RSNA 2019 in Chicago.
The Gemini 12 MP will be released in 2020 following clearance by the U.S. Food and Drug Administration (FDA). The monitor joins DBI's next-generation N Series and Gemini Series LED backlit display suite and will feature an ultrathin bezel, private light, higher luminance and contrast ratios, enhanced viewing of multimodality images, and integrated hybrid gamma correction.
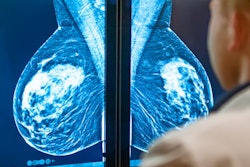
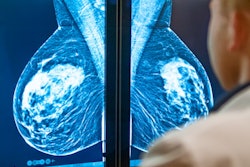
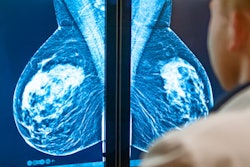
Displays include 3, 4, 5.2, 6, and 8.8 MP with the 5.2 MP and 8.8 MP displays FDA cleared for breast imaging, including tomosynthesis. The displays are also equipped with built-in front sensors for hands-free automatic DICOM 3.14 calibration, backlight sensors, and ambient light sensors to maintain stability and accuracy over time. The CFS calibration software suite for remote calibration, conformance, report generation, and nonconformance alerts via the web also is included with the displays.
Double Black is also highlighting its CFS productivity toolkit, which increases cursor movement efficiency while minimizing hand strain, fatigue, and frustration. The toolkit is adjustable by user and application and allows the user to concentrate on key areas of interest, automatically dim, and use the cursor effectively.