Breast imaging software developer Volpara Solutions plans to showcase its artificial intelligence (AI)-powered clinical-decision support platform for breast cancer screening and highlight two business partnerships at the RSNA 2019 meeting in Chicago.
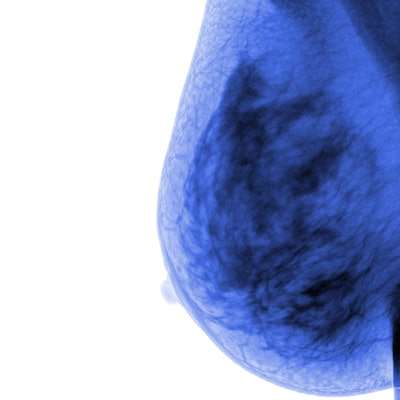
The newly redesigned VolparaScorecard+ software is designed to enhance radiologists' access to three key patient risk insights: breast density assessment, an indication of suspicious findings in the mammogram, and lifetime risk of developing breast cancer.
Volpara also will show its Volpara Live system, which automatically analyzes patient positioning and compression and provides immediate feedback to technologists on whether to retake images before a patient leaves the imaging session.
In other company news, Volpara Solutions is teaming with Dutch women's imaging software developer ScreenPoint Medical and MRS Systems, a provider of patient tracking, communication, and radiology reporting software for breast and lung imaging applications, to bring clinical decision-support tools to radiologists for screening for early detection of breast cancer.