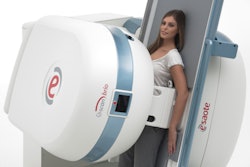
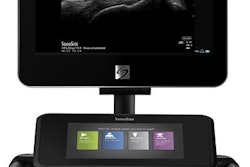
Ultrasound vendor Esaote has debuted its MyLab 9 eXP ultrasound system at RSNA 2017 in Chicago.
MyLab 9 eXP is specifically targeted to rheumatology, endocrinology, and prostate imaging, the firm said. It features single crystal noncomposite probe technology and appleprobe design to reduce musculoskeletal strain up to 70%, according to Esaote. The system also comes with image optimization and automation tools -- easyMode and easyTrace -- plus single-touch automated intelligent Doppler steering and angle correction.
Other features include the following:
- Virtual Navigator real-time Fusion, a multimodality application that is touch-screen based
- microV+, a noninvasive hemodynamic evaluator to characterize lesions
- QPack, a multimodality quantification tool of curve analysis of CnTI contrast-enhanced ultrasound perfusion, as well as color and power Doppler hemodynamics
- Solid-state hard disk, latest IntelCore i7 processor, and Windows 10
- Upgradable, remote service, long-term maintenance options, and transducer compatibility
MyLab 9 eXP is not currently for sale in the U.S. as it is awaiting clearance from the U.S. Food and Drug Administration.