CHICAGO - Radiation dose measurement is about to get a lot easier -- not to mention better and free of charge. Researchers from the National Cancer Institute (NCI) are finalizing a longstanding project to build a user-friendly software program that converts CT dose index volume (CTDIvol) scanner readouts into accurate organ dose estimates.
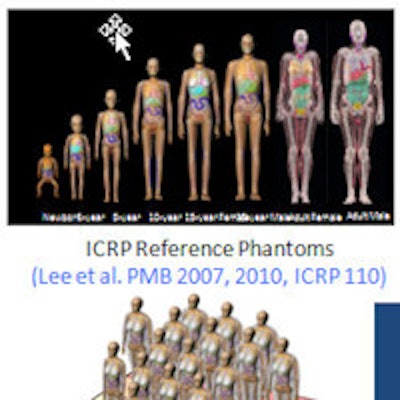
The just-published National Cancer Institute CT (NCICT) project shows the calculation for converting normalized CTDIvol to organ doses for six patient ages and both genders recently approved by the International Commission on Radiation Protection (ICRP), with software to be released early next year. An advanced version known as NCICTX, scheduled to be published next summer, will detail results calculated from 160 CT scanners and 170 size-dependent phantoms. Today, such conversions rely on much cruder tools.
"We have been working on our calculator for many years, and I wanted to get your comments," Choonsik Lee, PhD, from NCI told RSNA 2015 attendees.
The free computer program with graphical user interface (GUI) will enable radiologists to simply plug in the CTDIvol reading, along with protocol and patient parameters, to obtain organ dose estimates individualized to the full range of scanner models and patient sizes.
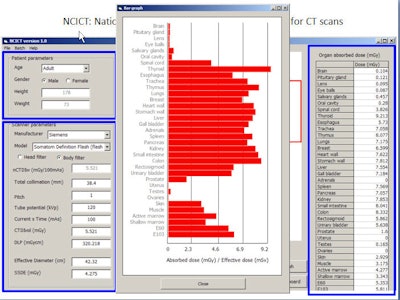
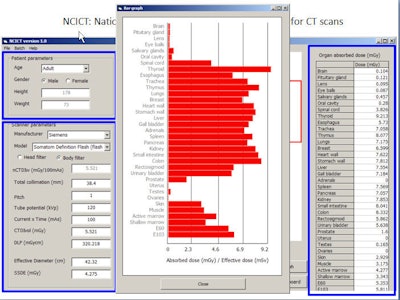
A user-friendly GUI will allow clinicians to input protocol scan parameters to quickly calculate organ dose, and the input does not need to be repeated when the same patient returns. All images courtesy of Choonsik Lee, PhD.
The software precalculates an organ dose library and displays the organ dose conversions based on the scan and patient particulars. The advanced version promises to be faster and more accurate than the NCICT program -- and light years ahead of current techniques, Lee said.
For example, when the investigators calculated 10 sample patient doses using both NCICT and a current dose calculation method, the CT-Expo technique, it revealed organ dose discrepancies of up to 200%.
"We compared organ doses from NCICT with organ doses from CT-Expo for a full-body scan and for pediatric phantoms," Lee said. "We found that some organs were in pretty good agreement, but some organs showed some significant differences between the NCICT and the current calculation method."
But despite NCICT's superiority to current methods, it's set for a major upgrade. The advanced NCICTX version of the software debuting next summer incorporates 170 new size-dependent phantoms, he said.
Labor-intensive task
There are two approaches to the mathematically challenging task of estimating organ doses, Lee explained. Manual measurements are reliable but also expensive and inflexible, and they require plenty of labor to produce. Automated calculation based on input criteria is fast and inexpensive, but it requires extensive validation before it can be used, which is the main task facing the NCI investigators.
The NCICT algorithm is based on work by Bahadori et al published this year in the American Journal of Roentgenology, and it incorporates a CTDI library based on machine output from dose measurements of more than 160 different scanners. Organ dose is then calculated from an organ dose library validated experimentally by Long et al. Based on this information, the library is being built and validated organ by organ.
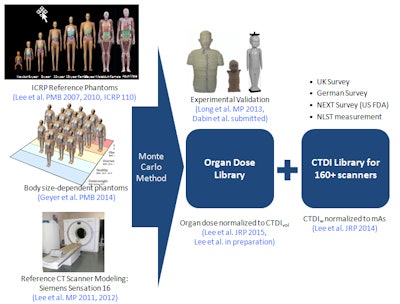
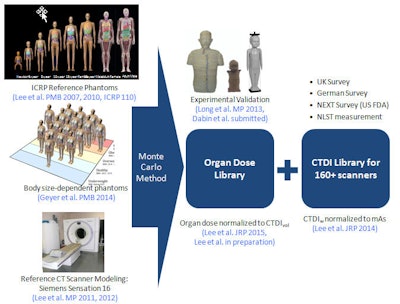
The NCICTX algorithm will incorporate advanced models for scanner output and patient sizes.
"If you can extract CTDIvol or slice-based mAs, you can use those parameters to calculate mA modulation and organ dose," Lee said of one study being prepared that compares lung dose calculations between NCICT and NCICTX. "We extracted height and weight from a screening trial and used that for the algorithm to calculate a region-specific lung dose based on NCICT and NCICTX."
In results to be published in 2016, the group found that NCICT, using reference size phantoms, overestimates the organ dose in patients with a high body mass index compared to NCICTX, Lee said.
NCICTX will deliver a broad range of improvements over existing methods of calculating organ dose, he added. While current retrospective dose reconstruction methods are based on a limited number of scanner models, NCICTX will be based on 160 models from eight manufacturers. Bone marrow dosimetry calculations will replace a model developed in the 1980s with a new ICRP model. Tube current modulation available in only a few samples will cover more scenarios. Finally, the new system will be available for free.
Besides releasing NCICT and NCICTX next year, the group is translating the results into Python for multiplatform use. Eventually, the application will estimate full organ-based tube current modulation as well, but "it's a lot of work," Lee told the audience.