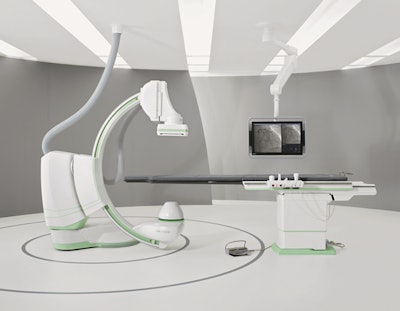
Siemens Healthcare is launching a new angiography system, called Artis One, designed to bring high-performance interventional imaging to the midrange segment.
Artis One targets what Siemens calls the expanding markets segment, where hospitals either don't have access to interventional imaging or are considering adding a third or fourth additional system. The unit was designed to perform standard procedures efficiently, without the specialized functions that might be available in a first or second angiography room, according to Francois Nolte, vice president of marketing and product management for Siemens' angiography and interventional x-ray segment.
That's reflected in the system's design, which features a small footprint that enables it to be sited in rooms as small as 25 sq meters, versus 45 sq meters for most angio systems. It also uses up to 20% less power than the previous generation of Artis, and the unit can be installed in 3.5 days, compared with a week previously. The unit also has a larger 30-inch display.
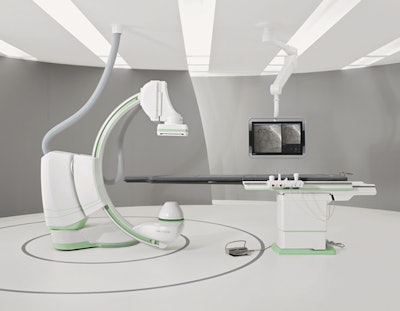 The new Artis One angiography system. Image courtesy of Siemens.
The new Artis One angiography system. Image courtesy of Siemens.But Siemens didn't cut corners in terms of important features such as image quality, Nolte said. In developing the system, the company used the same aspects of the imaging chain found on high-end systems such as the Artis Zee, including the detector, Megalix CAT Plus x-ray tube, and image processing chain.
Artis One also sports a new approach to table peristepping, a technique used when following a bolus injection down the periphery of a patient to see if there is a blockage in the legs. With most interventional x-ray systems, the table steps and the angiography unit remains stationary.
Instead, Artis One users can keep the table stationary and have the C-arm peristep down the table. The approach has benefits if clinicians have access lines running around the patient, enabling better patient access and capabilities not possible with a standard floor-mounted system.
Artis One will be offered with a floating tabletop, but it will not be available in a hybrid operating room (OR) configuration -- a Siemens customer would buy an Artis Zee or Artis Q for that, Nolte said. It might not be the best choice for a dedicated electrophysiology (EP) lab.
Siemens is in the process of filing a 510(k) application with the U.S. Food and Drug Administration (FDA) for Artis One, and the system will be highlighted at next week's RSNA 2013 meeting in Chicago. The company expects to be shipping internationally in the first half of 2014.