CHICAGO - A new CT scanner, a pair of new ultrasound systems, a wireless digital x-ray unit, and enhancements in coil technology for its MRI line are among the new product launches for Toshiba America Medical Systems at the RSNA 2011 meeting in Chicago.
CT
The new CT scanner is called Aquilion CXL, and the system sports a 64-detector-row, 128-slice configuration. Other technical specifications include a 7.5 million-heat-unit x-ray tube, 75-kW x-ray generator, and 0.35-sec gantry rotation time. Images can be reconstructed at a rate of up to 60 images per second.
The unit is pending 510(k) clearance from the U.S. Food and Drug Administration (FDA) and is being shown at RSNA 2011 as a work-in-progress.
Aquilion CXL also has the hardware required to support the latest generation of Toshiba's adaptive iterative dose reconstruction (AIDR) algorithm, called AIDR 3D. The software will be provided to Aquilion CXL users free of charge when it receives FDA clearance; AIDR 3D will also be provided for Aquilion One, Aquilion Premium, and Aquilion Prime scanners.
AIDR 3D is a major component of the new VeloCT console that Toshiba is highlighting at the meeting as a work-in-progress. The package also features faster reconstruction hardware and support for 128-slice scanning.
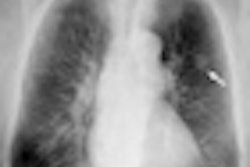
Making its U.S. debut in Chicago this week is Aquilion Prime, an 80-detector-row, 160-slice scanner that Toshiba launched at the European Congress of Radiology in March. Toshiba is touting the scanner's fast reconstruction speed with AIDR dose reduction, with reconstruction of up to 60 images per second. This system is also pending 510(k) clearance.
 |
| Toshiba's Aquilion Prime CT scanner is making its U.S. debut at RSNA 2011. |
In new clinical applications, Toshiba is showing two new functional imaging applications as works-in-progress: dual-energy imaging and myocardial perfusion. Dual-energy imaging uses two different energy levels to produce different information from a region of interest, with the scanner changing mA and kV levels during scanning.
On the advanced visualization side, Toshiba is demonstrating software from Vital, the 3D software developer that Toshiba acquired in June 2011. The company in particular is promoting Vital's Vitrea Enterprise Suite in its RSNA booth.
Ultrasound
In ultrasound, Toshiba is expanding its Aplio line with two new scanners: Aplio 300 and Aplio 500.
Aplio 300 is designed to be a workhorse scanner for routine ultrasound exams, including radiology, ob/gyn, and cardiac imaging. It includes clinical applications such as differential tissue harmonics (D-THI) and Toshiba's iStyle + productivity suite for better sonographer ergonomics.
 Aplio 300 is designed for routine ultrasound exams.
Aplio 300 is designed for routine ultrasound exams.Aplio 500 is a higher-end system designed to provide advanced visualization capabilities, better ergonomics, and workflow automation tools. It includes traditional clinical applications such as tissue-specific optimization (TSO), D-THI, iStyle +, and Advanced Dynamic Flow (ADF) technology, as well as two new Toshiba scanning protocols, Smart Fusion and Fly Thru.
Smart Fusion synchronizes CT images with ultrasound scans, enabling fused images to be displayed on a single screen. Toshiba believes the mode can help clinicians locate lesions and assist in ultrasound-guided biopsy procedures without the need for an additional CT scan.
Meanwhile, Fly Thru is an image reconstruction protocol that creates fly-through endoscopy-like 3D images from ultrasound data. Toshiba executives say the technique can be used for imaging any fluid-filled area of the body, and the company is demonstrating it in its booth with images of hepatic veins.
Aplio 500 also includes a number of cardiac applications migrated to the system from the company's premium Artida echocardiography scanner, making the new system appropriate for shared-services applications. New cardiac capabilities for Aplio 500 include 2D wall motion tracking and a new stress echo package, while Aplio 300 also has a stress echo package but without strain rate imaging.
Both new scanners have FDA clearance and are commercially available.
MRI
In the MRI section of its booth, Toshiba is displaying its 3-tesla Vantage Titan 3T MR scanner for the first time as a commercial product since receiving FDA clearance for the system in August. Toshiba has taken orders for 18 of the scanners and is currently installing the first two systems.
The company is also announcing a new series in the Vantage Titan line, Vantage Titan 1.5T MR, bringing the total number of systems in the product family to five. The new scanner adds 8-channel and 32-channel options to the existing 16-channel Vantage Titan, and Toshiba has also added two different coil options, standard coils and the new Helios coils.
 |
| Toshiba is displaying its Vantage Titan 3T MR scanner for the first time as a commercial product. |
The Helios coils offer higher slew rates than Toshiba's standard coils, at an amplitude of 34 mT/m and a slew rate of 203 mT/m/msec, compared with an amplitude of 33 mT/m and a slew rate of 148 mT/m/msec for the company's conventional coils. The Helios coils are pending FDA clearance and are being shown as works-in-progress; Toshiba is pointing out that customers can upgrade from 8-channel coils all the way up to 32 channels, and also from standard to Helios coils.
Also new in MRI is Octave, a new 8-channel gradient coil system, as well as new 16-element coils in a flexible rather than rigid design to improve versatility. Toshiba is demonstrating a configuration in which one of the flexible coils sits in a clamshell-shaped holder for pediatric brain scanning, while another coil lies flat on the patient table. The coils are available in small, medium, and large sizes, and they will be available for the company's 1.5-tesla and 3-tesla scanners when they receive FDA clearance.
Also new is a patient transport gurney that's designed to minimize handling for critically ill patients. Rather than move a patient from a conventional gurney to the MRI scanner's patient table, the new gurney simply slides over the top of the patient table, and the patient is scanned without having to be moved. The gurney, which is being shown as a work-in-progress, also includes slots for coils to make patient setup easier.
X-ray
 Kalare Wireless features a rotating bucky tray.
Kalare Wireless features a rotating bucky tray.New in the x-ray section of Toshiba's booth is Kalare Wireless, an update of the company's Kalare radiography room with a wireless digital radiography (DR) detector. A novel feature on the system is a rotating bucky tray, designed to minimize handling of the 14 x 17-inch detector by enabling technologists to reposition the detector from portrait to landscape orientation without removing it from the system. Kalare Wireless is commercially available.
In its interventional radiology line, Toshiba is promoting the flexibility of its Infinix line of angiography systems, with a special emphasis on productivity. Of particular note is a new 8-megapixel, 56-inch LCD monitor for the Infinix line, designed to make it easier for clinicians to see patient anatomy during interventional procedures. The monitor can change modes, either displaying one large fluoroscopy image or multiple display fields that might include both images and clinical information.