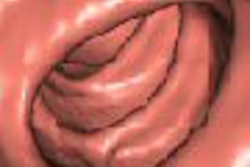
Forget 2D versus 3D. Creative types are working hard on new virtual colonoscopy visualization tools that go beyond simple fly-through endoscopic visualization, a technique that mimics the charming but limited view of the colonoscope.
Most of these so-called novel viewing methods are still in the early stages of development, while a few are all but ready for their close-up. All offer the potential of a powerful adjunct to 2D axial views, endoscopic fly-throughs, and CAD detection schemes for enhanced detection of polyps and cancer of the colon.
The potential payoff is better, more consistent reads of CT colonography datasets, with consistently higher sensitivity for significant lesions.
At the 2005 International Symposium on Multidetector-Row CT in San Francisco, Dr. Sandy Napel, a professor of radiology at Stanford University in Stanford, CA, discussed some of the interesting ideas he and his colleagues are exploring at Stanford and around the world.
Folds remain a critical limitation of the 3D endoscopic viewing, Napel said.
"Although these fly-throughs are wonderful, you have to fly in both directions to see as much behind the folds as you can," he said. "Even if you go in both directions, the folds tend to get in the way. The obvious problem is that folds are basically anatomic noise that impacts both visualization and CAD -- and that both humans and CAD algorithms are tuned to detect specific shapes."
Two ways to stretch
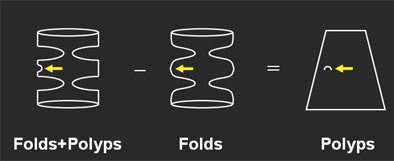
So why not get rid of them? A few researchers are doing just that, while others seek to visually enhance the lesions to stand out from folds, retained fluid, and stool.
David Paik, Ph.D.; Padma Sundaram; and colleagues from Stanford worked with Napel to develop an algorithm that tracks, then removes the folds, leaving the polyps behind for easy detection.
"(Paik's) idea is to model the colonic surface with a mathematical model that has very low frequency selectivity in the circumferential direction, but high-frequency connectivity in the (longitudinal) direction, so we wind up with a model that in effect tracks the fold, but does not track these small and midsize polyps," Napel said.
When the mathematical modeling is completed, the colon surface can be flattened or unrolled, "hopefully leaving the polyp behind on a flat sheet."
The resulting volumetric dataset can be further processed by CAD, Paik said in a presentation of the process at the 2004 RSNA meeting in Chicago. The group's colon phantom-based study concluded that fold height was reduced from 9.7 mm to less than 0.5 mm, while the polyps were extremely well-preserved. The successful results were repeated in a scan of a single patient.
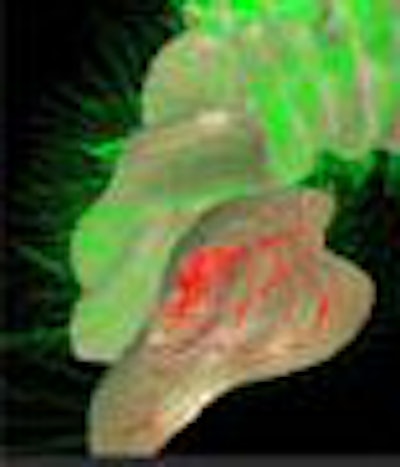
 |
| Geometric subtraction of anatomy is a process by which a low-order model of the colonic surface is created, then "subtracted" geometrically from the colon surface, rendering a flat surface on which polyps are still visible. Images courtesy of Dr. David Paik and Dr. Sandy Napel. |
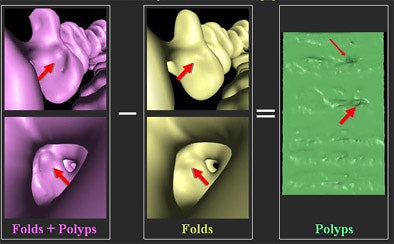 |
Another way to get rid of those pesky folds is to "stretch" the colonic mucosa like cloth to remove them. Napel said the idea came to him after seeing the work of a colleague in the computer science department, Ron Fedkiw, who develops "cloth" computer animation models for use in the film industry.
Fedkiw's team stretched out a physics-based simulation of cloth to get rid of the folds. Napel's associate Sundaram worked to adapt the concept to simulate stretching of the mucosa using a finite element model, with preliminary results -- also presented at RSNA 2004.
A level set of thresholded CT data was used to build a tetrahedral volumetric mesh representing the thickened colon wall, the team reported.
"We start from a surface mesh of the colon, and we ascribe physical properties to it, such that when we pull in opposite directions in a direction perpendicular to the folds, the folds will hopefully flatten out," Napel said. The team's results showed that surface curvature of the original unstretched folds (0.08, 0.12, 0.13) was flattened out completely in the resulting 3D volume.
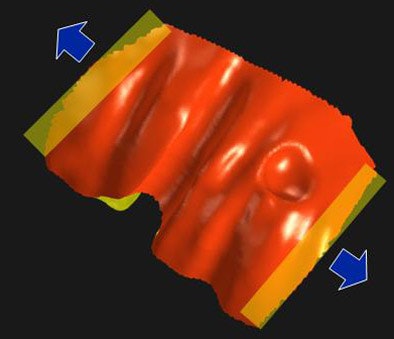 |
| In the finite element model, a level set of thresholded CT data is used to build a tetrahedral volumetric mesh representing the thickened colon wall. Various physical properties such as Young's modulus, Poisson's ratio, hardening deformation, and neo-hookean elasticity are attributed to the mesh, which is then subjected to stretching forces from two manually selected ends of the data, which erases the curvature of the folds. Image courtesy of Dr. Sandy Napel. |
"We have a ways to go, but this is a promising approach to try to reduce the appearance of folds," Napel said.
More obvious polyps
A similar result can be obtained by leaving the folds, but enhancing the appearance of the polyps. A former postdoctoral student at Stanford, Burak Acar, Ph.D., came up with the idea of using level sets to evolve the colonic surface to enhance the appearance of the sessile lesion, dubbed the polyp-enhancing level-set (PELS) method.
Acar continues his work on the PELS algorithms as part of the Volumetric Analysis and Visualization Group (VAVlab) in the electrical engineering department at Bogazici University in Turkey. The research team, which includes Ender Konukoglu, Dr. Christopher Beaulieu, Paik, and Napel, has submitted two related PELS algorithms for improving the performance of CAD algorithms for discussion at upcoming conferences.
One employs a nonlinear speed function, and will be presented at RSNA 2005 in Chicago. A second process, which Acar said he considers more mathematically robust than the first, has been submitted to the Medical Image Computing and Computer Assisted Interventions (MICCAI) conference for presentation later this month in Palm Springs, CA.
As for the first algorithm, "The underlying idea is to use the level-set-based interface (colon wall) propagation techniques to suppress the polyp necks while inflating the polyp apex," Acar and colleagues wrote in their RSNA 2005 abstract.
The enhancement and suppression is accomplished using an LS-based propagation along the surface normal direction according to a nonlinear speed function sensitive to the surface differential characteristics, the group stated.
In a polyp-rich CT dataset, when the speed-function-based PELS algorithm was added to the previously tested surface normal overlap (SNO) algorithm, the number of false-positive polyp detections was reduced from 107 to 24, while 100% sensitivity was retained for seven lesions 8 mm in diameter and larger, the group wrote in its abstract.
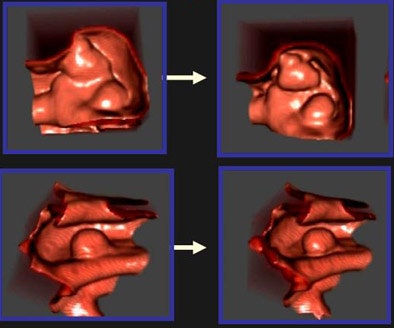 |
| The polyp-enhancing level-set (PELS) algorithm aims to improve the performance of both manual and CAD-based reading of CT datasets of the colon by altering their shape to enhance conspicuity. The PELS scheme uses a level-set (LS)-based interface to suppress the polyp neck while inflating the polyp apex, according to a nonlinear function of the surface principal curvatures for speed calculations. When applied to CT datasets in conjunction with a surface normal overlap scheme, the LS-based interface reduced false-positive markings fivefold. Image courtesy of Burak Acar, Ph.D. |
Beyond attenuation
A number of groups are working on processes that perform volume rendering through tagged fluid, Napel said.
Volume rendering uses an opacity function dependent on CT attenuation (Hounsfield units). Typical implementations of this function for virtual colonoscopy yield clearly visible anatomy above the fluid, but leave the tagged fluid completely opaque, obscuring the view of the colonic wall below the fluid. New efforts are focused on incorporating other imaging properties, such as image gradients and texture, which can allow transparent rendering of the fluid near the air-fluid interface, thereby enabling visualization of soft tissues below the fluid. The method might even turn out to be superior to digital subtraction, as it does not run the risk of losing surface features in the subtraction process, Napel said.
Someday, when CAD algorithms are more advanced, reading studies could get a whole lot easier, he said. One possibility is a graphic user interface that presents volume-rendered images of, say, the top 16 CAD results. Clicking on one generates an automated fly-through, allowing the finding to be called as a true or false positive very quickly.
"We've talked about preprocessing for better visualization, getting rid of folds, enhancing polyps, and I think these are going to go a long way toward helping human interpretation as well as CAD," Napel said. "We need more flexible volume rendering so that we don't necessarily have to subtract tagged fluid. User interfaces are certainly under development, and will evolve into the future, as well as integration with other data and databases."
By Eric Barnes
AuntMinnie.com staff writer
October 13, 2005
Related Reading
'Filet view' VC software slices reading time, October 6, 2005
VC path-planning technique maximizes observable mucosa, August 30, 2005
Colon CAD: VC's extra eyes face new challenges, August 5, 2005
Part II: Computer-aided detection identifying new targets, July 15, 2005
Virtual colon dissection is promising, if flawed, October 9, 2003
Copyright © 2005 AuntMinnie.com