Oncologists and nuclear medicine technologists may soon have another weapon in their arsenal to diagnose and evaluate various cancers through PET/CT, according to a study published in the June issue of the Journal of Nuclear Medicine.
German researchers have developed a class of radiopharmaceuticals based on gallium-68 (Ga-68) that acts as fibroblast activation protein inhibitors (FAPIs). Thus far, the radiotracers have proven effective in identifying nearly 30 types of malignant tumors with greater uptake and improved image contrast (JNM, June 2019, Vol. 60:6, pp. 801-805).
"Several epidemiologically important tumor entities, in particular breast, esophagus, lung, pancreatic, head-neck, and colorectal cancer, present with a remarkably high uptake in Ga-68 FAPI PET/CT," wrote the researchers led by Dr. Clemens Kratochwil from University Hospital Heidelberg. "This may open new applications for noninvasive tumor characterization and staging examinations."
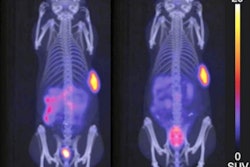
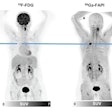
The key is the Ga-68 FAPI radiotracer's ability to target fibroblasts, which can account for as much as 90% percent of a tumor's mass and promote tumor growth, migration, and progression. In a March 2019 proof-of-concept study, the researchers showed how Ga-68 FAPI-2 and Ga-68 FAPI-4 provide high-contrast images with quality equal to or better than that of the standard FDG-PET/CT imaging.
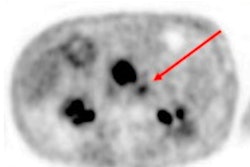
For this retrospective study, Kratochwil and colleagues scanned 80 patients with 28 different kinds of cancer using a commercially available PET/CT scanner (Biograph mCT, Siemens Healthineers). The cancers in the patient population included 229 histopathologically proven metastases and 54 primary tumors. Ga-68 FAPI administrations ranged from 122 to 312 MBq. Tracer uptake was measured by mean standardized uptake values (SUVmean) and maximum SUV (SUVmax) one hour after injection.
The researchers observed the highest average SUVmax (greater than 12) in cases of sarcoma, esophageal, breast, cholangiocarcinoma, and lung cancer. The lowest Ga-68 FAPI uptake (average SUVmax of less than 6) was seen in pheochromocytoma, renal cell, differentiated thyroid, adenoid cystic and gastric cancers. The average SUVmax of hepatocellular, colorectal, head-neck, ovarian, pancreatic, and prostate cancer was intermediate (SUVmax range, 6-12).
"All patients tolerated the examination well," the researchers noted, adding that "no drug-related pharmacologic effects or physiologic responses occurred."
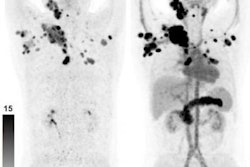
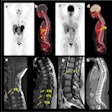
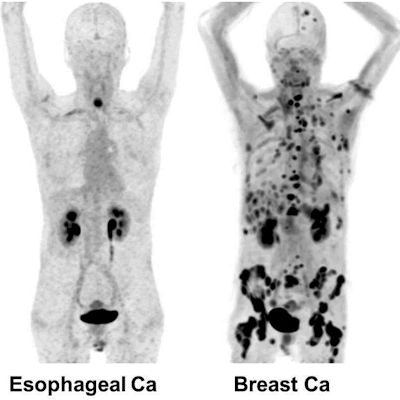
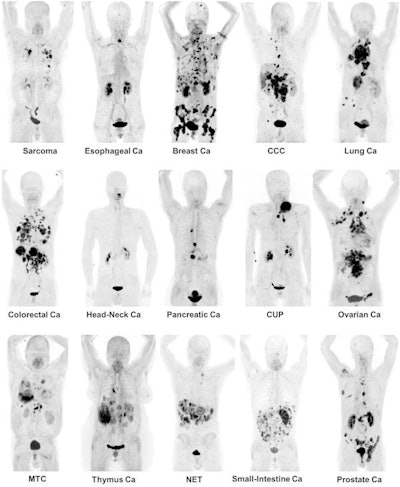 Maximum-intensity projection images of Ga-68 FAPI PET/CT in patients with 15 different histologically proven tumor entities (sorted by uptake in descending order). Ca = cancer; CCC = cholangiocellular carcinoma; CUP = carcinoma of unknown primary; MTC = medullary thyroid cancer; NET = neuroendocrine tumor. Images courtesy of Kratochwil et al and JNM.
Maximum-intensity projection images of Ga-68 FAPI PET/CT in patients with 15 different histologically proven tumor entities (sorted by uptake in descending order). Ca = cancer; CCC = cholangiocellular carcinoma; CUP = carcinoma of unknown primary; MTC = medullary thyroid cancer; NET = neuroendocrine tumor. Images courtesy of Kratochwil et al and JNM."The remarkably high uptake of Ga-68 FAPI makes it useful for many cancer types, especially in cases where traditional F-18 FDG PET/CT faces limitations," added co-author Dr. Uwe Haberkorn, from the University Hospital of Heidelberg and the German Cancer Research Center, in a statement. "For example, low-grade sarcomas generally have a low uptake of F-18 FDG PET/CT, causing an overlap between benign and malignant lesions."
Another benefit to patients is that Ga-68 FAPI PET/CT can be performed with no specific patient preparation, such as fasting. In addition, the researchers see the Ga-68 FAPI approach as a potential tool for theranostic radiopharmaceuticals that can deliver treatment in the future.
"Because the Ga-68 FAPI tracers contain the universal DOTA-chelator, it is possible to label them with therapeutic radionuclides whose half-life fits to the tumor retention time of the carrier molecule," Haberkorn added. "Since the tracer has been observed to accumulate in several important tumor entities, there may be a huge field of therapeutic application to be evaluated in the future."