A deep-learning algorithm called HeadXNet can improve the performance of clinicians of varied specialties and levels of experience for detecting intracranial aneurysms on head CT angiography (CTA) exams, according to research published online June 7 in JAMA Network Open.
A team of researchers from Stanford University, led by Allison Park, trained its segmentation model based on deep learning to provide voxel-by-voxel predictions of intracranial aneurysms on CTA. In testing, eight clinicians -- including a surgeon, a radiology resident, and six board-certified radiologists -- experienced a statistically significant increase in sensitivity, accuracy, and interrater agreement. These gains came at no cost to specificity or diagnosis time.
"This suggests that integration of an artificial intelligence-assisted diagnostic model may augment clinician performance with dependable and accurate predictions and thereby optimize patient care," the authors wrote.
CTA is the primary minimally invasive imaging modality for diagnosis, surveillance, and presurgical planning for intracranial aneurysms, which account for more than 80% of nontraumatic life-threatening subarachnoid hemorrhages. However, interpretation of these studies is time-consuming even for subspecialty-trained neuroradiologists and is subject to low interrater agreement, according to the researchers.
"The development of an accurate deep-learning model to help clinicians reliably identify clinically significant aneurysms in CTA has the potential to provide radiologists, neurosurgeons, and other clinicians an easily accessible and immediately applicable diagnostic support tool," the authors wrote.
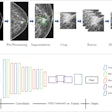
The researchers utilized a 3D convolutional neural network that was trained on a set of 611 head CTA studies to produce aneurysm segmentations. The researchers then provided the eight clinicians with the segmentation outputs from a test set of 115 exams. Over two sessions separated by 14 days, the clinicians then read the exams in randomized order either with or without model augmentation as part of a crossover design.
Use of HeadXNet led to a significant improvement in sensitivity, accuracy, and interrater variability.
| Mean clinician performance for detecting intracranial aneurysms | ||
| Without HeadXNet | With HeadXNet | |
| Sensitivity | 83.1% | 89%* |
| Specificity | 96% | 97.5% |
| Accuracy | 89.3% | 93.2%* |
| Interrater agreement | Exact Fleiss kappa = 0.799 | Exact Fleiss kappa = 0.859* |
What's more, it did not lead to a statistically significant increase in mean diagnosis time per examination.
"In addition to significantly improving accuracy across clinicians while interpreting CTA examinations, an automated aneurysm detection tool, such as the one presented in this study, could also be used to prioritize workflow so that those examinations more likely to be positive could receive timely expert review, potentially leading to a shorter time to treatment and more favorable outcome," the authors wrote.
The significant improvement in sensitivity, accuracy, and interrater agreement implies that the model could lead to more accurate and more consistent radiological interpretations, according to the researchers. They said that future work on the model should include prospective evaluation and testing on imaging data from other institutions and hospitals.