PET scans with the novel radiotracer flortaucipir may provide more valuable information in the early detection of neurodegeneration associated with Alzheimer's disease than MRI, according to a study published in the February issue of the Journal of Nuclear Medicine.
F-18 flortaucipir is designed to bind to amyloid and tau-based neurofibrillary tangles (NFTs), which influence the decline of Alzheimer's patients who have memory loss or physical deficiencies from the disease. For the latter group of patients, their limitations from Alzheimer's often occur earlier than more typical signs of impairment.
"These groups provide an ideal context to test whether [F-18 flortaucipir] can detect known regional differences and investigate associations between NFTs and other measures of neurodegeneration," wrote lead author Dr. Ilya Nasrallah and colleagues from the University of Pennsylvania. "With this and other recent studies showing strong correlations between tau PET and imaging measures of neurodegeneration, it will be critical to establish the role of tau PET with respect to other biomarkers of Alzheimer's disease pathology."
Flortaucipir research
Mild cognitive impairment occurs in approximately 14% to 18% of people older than 70, according to the Alzheimer's Association. The most common form of the condition includes memory loss; such patients are considered amnestic. Less common are the 5% to 6% of seniors who have Alzheimer's and are nonamnestic. They show no signs of memory deficiency, but they often have difficulty speaking, which becomes progressively worse.
Flortaucipir, also known as F-18 AV-1451 and F-18 T807, is being developed by Avid Radiopharmaceuticals. Over the past few years, the tracer has shown a propensity for targeting tau protein accumulation in the brain in preclinical and early clinical studies.
Researchers from Washington University in St. Louis used flortaucipir in 2016 to evaluate neurofibrillary tau pathology in vivo. They found that flortaucipir-PET images correlated with regionally specific cortical thinning in Alzheimer's disease.
In a September 2016 study, researchers from Mount Sinai in New York City found that flortaucipir was effective for highlighting tau protein in relation to diagnosing concussion-related brain degeneration for the first time in a live subject. The findings were particularly encouraging because tau has been linked to chronic traumatic encephalopathy (CTE), which so far can only be confirmed at autopsy.
For the current study, the researchers prospectively recruited patients in 2015 with an onset of cognitive decline prior to age 65. Six subjects were amnestic with early-onset Alzheimer's and presented with posterior cortical atrophy, while four subjects had progressive aphasia and were nonamnestic. The study also included six age-matched Alzheimer's patients and six control subjects.
PET brain scans (Ingenuity TF, Philips Healthcare) were performed 80 to 100 minutes after injection of 370 MBq of flortaucipir. PET images were generated by iterative reconstruction with CT attenuation correction to evaluate the tracer's uptake and its ability to bind to tau protein. In addition, subjects underwent 3-tesla MRI scans with a volumetric T1-weighted protocol to measure gray-matter atrophy (JNM, February 2018, Vol. 59:2, pp. 299-305).
With the addition of regional and voxel-based morphometry, Nasrallah and colleagues assessed flortaucipir binding and gray-matter density through the T1-weighted MR images and standardized uptake values (SUVs) from PET. Cognitive tests to assess memory and language skills were performed at a mean of eight days (range, 0-32 days) after the flortaucipir-PET scans.
Voxel-based analysis
Based on the voxel-based morphometry of SUV and gray-matter density, MRI was not as proficient in differentiating between Alzheimer's patients and controls than flortaucipir-PET.
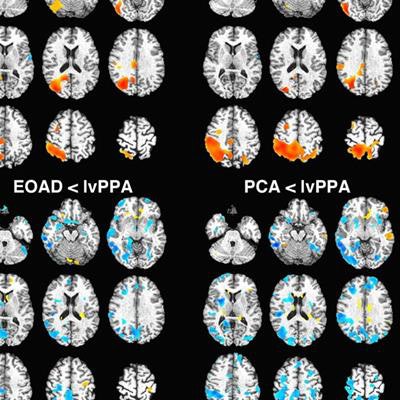
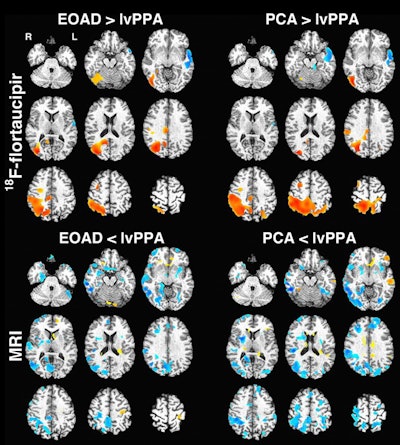 Voxel-based images show comparison of flortaucipir-based standardized uptake values (SUV) (top) and gray-matter density (bottom). Regions with higher SUV or gray-matter density in early-onset Alzheimer's disease (EOAD)/posterior cortical atrophy (PCA) versus lvPPA (logopenic variant of primary progressive aphasia) are in red-yellow, with those higher in lvPPA in blue. There was no significant difference between early-onset Alzheimer's and posterior cortical atrophy groups for PET or MRI. Images courtesy of JNM.
Voxel-based images show comparison of flortaucipir-based standardized uptake values (SUV) (top) and gray-matter density (bottom). Regions with higher SUV or gray-matter density in early-onset Alzheimer's disease (EOAD)/posterior cortical atrophy (PCA) versus lvPPA (logopenic variant of primary progressive aphasia) are in red-yellow, with those higher in lvPPA in blue. There was no significant difference between early-onset Alzheimer's and posterior cortical atrophy groups for PET or MRI. Images courtesy of JNM.As one might expect, flortaucipir binding in nearby regions-of-interest within the Alzheimer's groups compared with controls (p < 0.05). The most prominent differences were in the right and left temporoparietal and medial temporal regions (p < 0.001).
The posterior cortical atrophy group also showed the highest radiotracer binding in the medial parietal/posterior cingulate and lateral occipital lobe (p < 0.001), as well as the right and left temporoparietal, medial temporal, and medial parietal/posterior cingulate regions (p < 0.001) in the control group comparisons.
Given flortaucipir's ability to bind to the tau deposits and the association with neurodegeneration, the researchers believe the radiotracer can be "critical" to establishing the tau-PET connection to other biomarkers of Alzheimer's pathology, including beta amyloid, regional atrophy, and cerebrospinal fluid.
The author cited several limitations to the study, including the small number of subjects, which could affect the assessment of differences between the groups. Also, they noted that voxel-wise comparisons between Alzheimer's groups will require additional validation.