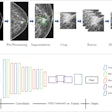
Image analysis software developer Quantitative Insights has received U.S. Food and Drug Administration (FDA) clearance for QuantX Advanced, a breast imaging computer-aided diagnosis platform that utilizes artificial intelligence (AI) technology.
Based on machine learning and analytics, QuantX features tools such as QI Score and Similar Case Database Compare, which give users access to a database of breast abnormalities for comparison with known pathology, the company said. The software includes technology licensed from the research labs of Maryellen Giger, PhD, of the University of Chicago. She serves as a scientific advisor for the firm.
Quantitative Insights is now seeking distribution partnerships for both the U.S. and internationally.