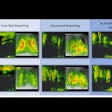
Canadian radiography quality control technology developer Diagnostic Imaging Specialists (DISC) of St. Malo, Manitoba, plans to launch its new DR Radchex device at the RSNA meeting later this month.
DR Radchex is designed for weekly and periodic digital radiography (DR) quality assurance programs on DR systems from all vendors. It features an 11.5 x 11.5-inch detector located in the center of its electronic cassette to allow imaging phantoms to be used to create images, while simultaneously providing a measurement of radiation dose used.
Related Reading
DISC adds distributor, August 17, 2010
DISC inks deal with Broadwest, May 3, 2010
Diagnostic Imaging Specialists signs Pyers, February 11, 2010
Copyright © 2010 AuntMinnie.com