CHICAGO - New developments in CT dose reduction, an expansion of its digital MRI architecture, and new enhancements in PET/MRI are pacing product news in the RSNA 2012 booth of Philips Healthcare.
CT
Radiation dose reduction has become a hot topic in CT, and Philips is demonstrating the latest iteration of its iterative reconstruction algorithm, iDose4 Premium Package.
Philips is highlighting the reliability, image quality, and low dose levels possible with iDose4 Premium Package, with some CT procedures producing dose levels as low as 1 mSv. A major component of the algorithm is its metal artifact reduction for large orthopedic implants (O-MAR), which reduces artifacts caused by these devices on CT scans.
iDose4 Premium Package is available on the iCT and Ingenuity CT scanner families, as well as Brilliance CT 64-channel scanners.
Philips is also showing work on its next dose-reduction technology, iterative model reconstruction (IMR). Philips is calling IMR a "knowledge-based reconstruction" technique that has the ability to produce "noise-free" images, with a nearly threefold improvement in low-contrast detectability, which could be particularly useful to visualize lung parenchyma in large patients, for example.
IMR's advanced reconstruction capabilities require more computer processing power, but Philips engineers have found a way to make more rapid reconstruction possible, with reconstruction times as low as five minutes for IMR images.
Philips is showing IMR as a commercial product in its RSNA booth, and the company will offer it as a product to its installed base of iCT users, with deliveries expected in the second half of 2013. Philips is also making the older iDose algorithm available as part of the standard configuration for its entire CT installed base.
Another new technology is iPatient, designed to help users better manage image quality, radiation dose, and contrast injector protocols. The concept includes SyncRight, which helps users develop contrast injection protocols that are personalized to the patient being scanned, and that can be repeated consistently.
SyncRight can be set so that only the contrast parameters that need to be adjusted by the operator are visible (although users can also opt to see all parameters available, if necessary). Also, scanning protocols can be shared with other users via Philips' ExamCards network.
In a future technologies section of the company's booth, Philips is showing what it calls spectral detection CT (SD CT). The work-in-progress technology will potentially use detectors made from yttrium rather than gadolinium oxysulfide (GOS). Yttrium detectors are expected to have better light acquisition properties, and would give clinicians the ability to visualize spectral images at the same time as routine CT data without having to perform a separate scan.
Molecular imaging
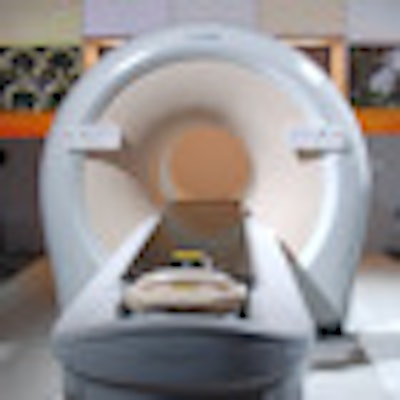
In the molecular imaging section of its RSNA booth, Philips is promoting Ingenuity TF PET/MRI, the hybrid scanner that the vendor introduced in 2011.
 |
| Philips' Ingenuity TF PET/MRI with MultiTransmit. |
Among recent enhancements to the system are improvements in the scanner's time-of-flight (TOF) algorithm, now on the fourth generation of TOF technology. The new version results in sharper images and also dramatically accelerates image processing speed, with images available just a few minutes after they are acquired. TOF-based PET is now available across the entire Philips PET portfolio.
Also new on the PET/MR scanner is the same MultiTransmit parallel-imaging technology found on the company's flagship Achieva 3.0 TX MR scanner. MultiTransmit MRI uses radiofrequency (RF) coils in which the frequency, amplitude, phase, and waveform of all RF sources are automatically adjusted for optimal uniformity for each patient's anatomy; it enables the PET/MRI scanner to achieve image acquisition speeds comparable to PET/CT.
For the company's Ingenuity PET/CT scanner, Philips is showing all the new technologies that have been introduced on the CT line, such as the iPatient protocol management system and iDose4 Premium Package with O-MAR.
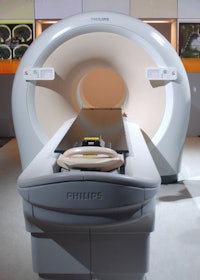 Philips' Ingenuity scanner.
Philips' Ingenuity scanner.In other RSNA news, Philips is bringing its full suite of nuclear medicine applications into its IntelliSpace Portal workstation environment, which previously only supported CT and MR images. IntelliSpace Portal applications are accessible virtually anywhere on PACS workstations or PCs, and nuclear medicine images can now be reviewed next to CT and MRI scans.
Finally, Philips is extending its Ambient Experience concept into radiopharmaceutical injections. The company is showing a special room designed for patients receiving FDG injections, who have to wait one hour between when they are injected and when they can be scanned.
This can be a stressful time, so the Ambient Experience room allows them to pass the time in a custom environment with nature scenes and soothing music. Developed in cooperation with the Dutch Cancer Institute, the design has been shown in a clinical study to reduce patient anxiety, which could lead to more accurate exams. The rooms are expected to be commercially available in mid-2013.
MRI
Philips is demonstrating a range of new MRI technology at RSNA 2012, including a new version of its Ingenia platform optimized for the operating room, a mobile version of Ingenia, the expansion of its digital RF technology to more scanners, and a new scanner targeted at the economy segment.
First up is Ingenia MR-OR, a new system that combines a surgical suite with the company's Ingenia scanner. The configuration is designed to enable the rapid transfer of patients into the MRI bore, which Philips believes can reduce the need for invasive surgery.
For example, the scanner can help brain surgeons visualize procedures in real-time, allowing immediate access to patient information that could affect surgical decisions. Ingenia MR-OR is available at 1.5- and 3-tesla field strengths.
Ingenia Mobile is a mobile version of the Ingenia platform. It's currently available at 1.5 tesla, but Philips is also working on a 3-tesla version.
On the scanner instrumentation side, Philips is rolling out its SmartPath to dStream upgrade, which brings the dStream digital broadband technology to other MRI scanners in the Philips product line. dStream digitizes signals inside the RF coil, resulting in up to a 40% improvement in signal-to-noise ratio.
dStream also offers economic benefits in addition to improved performance, according to the company. Users have seen throughput increases of up to 30%, and those who upgrade to the technology have lower costs when upgrading to new RF channel configurations.
 |
| Philips is touting its Ingenia MR-OR system. |
On the interventional side, Sonalleve MR-HIFU (high-intensity focused ultrasound) is a module for Ingenia scanners that enables them to perform noninvasive HIFU treatment for conditions such as uterine fibroids and adenomyosis, as well as palliative care of bone metastasis. The system is not yet available for sale in the U.S. and is being demonstrated at RSNA as an investigational device.
Finally, Multiva is a new 1.5-tesla scanner first shown at the Journées Françaises de Radiologie (JFR) congress in Paris in October. The system is designed for routine applications for facilities with stricter budget requirements, and it sports lower coil-handling time for better workflow.
Multiva will carry a price point about 30% less than an Ingenia 1.5-tesla scanner; it's being rolled out in some geographic markets now, with availability in North America in the second half of 2013. It is not available for sale in the U.S. and is being shown as a work-in-progress.
Interventional radiology
In the interventional section of its booth, Philips is highlighting products designed to support the move in healthcare away from open surgical procedures. One such product is a hybrid operating room (OR) with FlexMove, a technology that fuses an x-ray angiography room with an OR to give physicians the best of both worlds.
With FlexMove, the angiography system can be parked in a corner of the OR and brought in when needed to offer imaging support during the surgical procedure. FlexMove began shipping at the end of 2011.
In interventional oncology, Philips is developing solutions around invasive procedures for visualizing vasculature around tumors. The company has signed a comarketing deal with medical technology firm BioCompatibles that includes training courses on how to use each firm's technology together for interventional oncology.
The company is also showing the integration of new imaging techniques on angiography. XperCT offers CT-like images on the angiography unit, while a Philips CX50 ultrasound scanner can be added to the angio suite to provide additional guidance information; data can be imported into a PACS and stored next to x-ray information for easier access.
Finally, in interventional neuroradiology, Philips will show XperCT with VasoCT, an advanced protocol for supporting stroke intervention. The technique offers high-resolution imaging of "pipeline" stents for positioning, as well as imaging of occlusive strokes to determine extent of vessel blockage.
X-ray
Wireless portable detector (WPD) sharing for digital radiography (DR) detectors is the major focus in the x-ray section of Philips' RSNA booth.
Philips wireless portable detectors can now be used across all digital x-ray systems in the company's product line, from mobile units to chest stands to x-ray tables. Philips customers can now use one to two detectors in multiple x-ray rooms, or they can use a wireless detector on rounds in the wards in the morning and in fluoroscopy rooms in the afternoon.
 |
| Christina Textor from Philips shows the wireless digital detector. |
The company is providing upgrades from existing systems to WPD sharing, with the kits consisting of a software upgrade and the addition of a portable detector.
Meanwhile, DuraDiagnost is a new customizable DR room designed for geographic markets outside the U.S. It's designed to offer DR technology at half the cost of competing systems. It will be available in a single-detector configuration with the detector in a bucky, or in a dual-detector setup that includes a wall stand. All DuraDiagnost rooms are floor-mounted.
Philips is also marking the 20th year of its digital x-ray technology, with more than 6,500 systems installed worldwide.
Women's imaging
In the women's imaging section of its booth, Philips is demonstrating the MicroDose mammography line, which the company gained through its acquisition of Sectra's mammography business in 2011.
The big news at RSNA 2012 is MicroDose SI, a single-shot spectral imaging technology that provides spectral breast density measurements. Like other MicroDose systems that are commercially available, MicroDose SI uses direct, digital photon-counting technology. Awareness has been growing of the relationship between breast density and cancer risk, and MicroDose SI offers users the ability to produce quantitative density measurements from mammography images.
MicroDose SI generates measurements that can be repeated in subsequent exams, and can help guide the use of additional imaging modalities that might be preferable to mammography for women with dense breasts. The technology is pending 510(k) clearance from the U.S. Food and Drug Administration (FDA) and is being shown as a work-in-progress. It is expected to be commercially available in 2013.
The company is also showing advanced applications on its IntelliSpace Breast workstation, a multimodality workstation with the ability to display mammography, digital tomosynthesis, breast MRI, and breast ultrasound images. It now has the ability to support breast density assessment, integrating data directly into BI-RADS standard report templates, as well as a number of automated reporting tools to facilitate workflow.
Both MicroDose SI and the IntelliSpace Breast workstation were displayed in a space showing Ambient Experience room enhancements. As competition increases in the women's imaging market segment, Philips said there has been growing demand by imaging centers and radiology departments to differentiate the experience of having a mammogram with an environment designed to reduce anxiety. Customers have reported that Ambient Experience also enhances the work experience of mammographers.
Finally, Philips is also demonstrating a new silicon-based detector that will be the company's mammography platform of the future, although it still uses the MicroDose photon-counting technology. International shipments are scheduled to begin in January.
Ultrasound
There's a lot going on in the ultrasound zone of Philips' booth this year. Elastography imaging is a major theme, as are new surgical transducers, and a bevy of new features for its premium iU22 xMatrix ultrasound system.
Philips is highlighting elastography technology, which measures tissue stiffness as a possible indicator of pathology, on its iU22 xMatrix scanner. The company is building on research that suggests elastography measurements can indicate whether breast tissue is benign or malignant -- information that can help prevent unnecessary biopsies. The firm is looking to expand elastography's role with new applications in breast imaging, and also new probes in areas such as gynecology and liver imaging (being shown as works-in-progress).
The company is also moving beyond traditional tissue strain-based elastography and into shear-wave elastography. Philips has been marketing shear-wave elastography outside the U.S. and is showing it at RSNA 2012 as a work-in-progress, pending FDA clearance.
New features for the iU22 xMatrix system include new anatomical intelligence tools that use a database of anatomical structural models with adaptive system intelligence designed to simply exams, provide easy acquisition, and improve reproducibility. These include a fetal heart navigator and vascular plaque quantification.
When the first portable scanner was introduced several years ago, it was intended to make performing ultrasound exams in intensive care units and neonatal intensive care units easier. While customers liked the portability, they also wanted a portable compact system that offered higher-quality imaging.
Philips' response was the CX50 CompactXtreme scanner, which it is highlighting at the meeting for pediatric use. Also being highlighted are three new surgical transducers for the scanner, designed for vascular surgery (L15-7io), for renal and hepatic laparoscopic surgery (L10-4lap), and for imaging deep within incisions such as those needed for liver surgery (C0-3io). All of these transducers were displayed as works-in-progress for North America; they are available commercially in Europe, Asia, and most other global markets.
Finally, Philips is also migrating its PureWave technology from iU22 xMatrix to the HD15 scanner, available both in the U.S. and internationally. PureWave's C5-1 transducer is being highlighted this year for use with obese patients.
Advanced visualization
On the advanced visualization side, IntelliSpace Portal is the featured technology. The workstation platform was first introduced at the 2010 RSNA meeting and began shipping in 2011.
 Lou Bonincontri from Philips demonstrates the intelliSpace Portal workstation.
Lou Bonincontri from Philips demonstrates the intelliSpace Portal workstation.The new version of the platform, IntelliSpace Portal v5.0, brings nuclear medicine applications into the environment, which already supports CT and MR images. Philips is highlighting the cloud-based design of the platform, which enables images to be securely transferred and accessed from virtually any location, as well as its "zero-click" functionality for automated image processing, which boosts throughput by up to 80%.
The first installations of v5.0 are occurring in November and December, with full deliveries in the first and second quarter of 2013.
PACS
Philips' imaging informatics portfolio is focusing on the very small and the very large. Its new IntelliSpace PACS DCX software is being promoted as an advanced enterprise PACS with workflow-oriented image management over many geographic boundaries.
The software is designed for customers with traditional PACS requirements to simplify workflow, support clinical decision-making, and improve financial outcomes through increased efficiency and interoperability. It is licensed with a flexible application model to accommodate single-modality imaging centers through large enterprises.
IntelliSpace PACS Anywhere, shown as a work-in-progress, is a zero-footprint image and report viewer fully integrated into IntelliSpace PACS 4.4. It is being designed for use on multiple platforms, including a wide variety of mobile devices.