Siemens Healthcare is planning to launch a new line of premium angiography systems, called Artis Q, at the upcoming RSNA meeting in Chicago. The new line includes enhancements in detector design, guidance technology, and x-ray tube instrumentation.
The new systems will come in two configurations, Artis Q and Artis Q.zen, with the latter system occupying the flagship position in the vendor's angiography portfolio. Both systems will carry new Siemens x-ray tube technology designed to reduce dose, while Artis Q.zen will sport both redesigned digital detectors as well as new visualization techniques for procedures such as stent placement.
At the heart of the new line is the second generation of Gigalix flat-emitter x-ray tube technology that Siemens has developed over the past five to six years. Instead of the coiled filaments used in conventional tubes, flat emitters built around tightly machined pieces of tungsten are used to emit electrons.
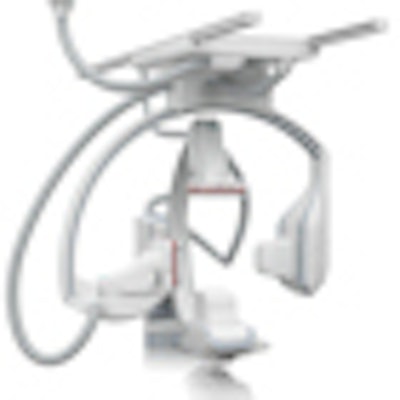
 Artis Q.zen. The red stripe denotes the system's crystalline silicon detectors.
Artis Q.zen. The red stripe denotes the system's crystalline silicon detectors.Siemens compares the leap to flat-emitter technology to be similar to the difference between incandescent light bulbs and light-emitting diodes (LEDs), according to Jim Williams, vice president of marketing and product management for angiography and interventional radiology. The upshot of the technology is better visualization of small structures typically encountered in interventional radiology.
"We can pump more current through the tube faster, with a smaller focal spot," he said in an interview with AuntMinnie.com. "A small focal spot helps with spatial resolution -- we can see really tiny guidewires."
Meanwhile, Artis Q.zen also features new digital detector technology that's based on crystalline silicon rather than the amorphous silicon previously used. The crystalline structure is more homogenous and thus allows for more effective amplification of signals.
That translates into lower radiation dose, according to Williams. In field tests at sites in Europe, the system is producing fluoroscopy doses that are sometimes half of what might be found with a conventional amorphous silicon detector, he said.
"This is a big breakthrough for us, especially where you are concerned about long procedures, where the patient may be on the table for three to six hours, and you need to fluoro for a long time," he said.
Finally, Siemens has developed new interventional applications that it hopes will match the advances made on the hardware side for Artis Q.zen. One application for cardiology guidance is called syngo CLEARstent Live: it recognizes and tracks stents in dynamic cardiac images, correcting for motion and using an overlay to create a significantly enhanced image of the stent, Williams said.
Basically, CLEARstent Live compensates for motion so the interventional physician has the impression of a perfectly motionless stent, according to Williams. The physician can then thread a guidewire through the stent more easily. The application is particularly useful for placing stents one after another.
Another new application is syngo DynaCT Micro, an enhanced version of Siemens' DynaCT tool for performing CT-like imaging on an angiography system. DynaCT Micro uses the full resolution of the detector for the technique, and also is now using 16 bits of native resolution rather than 14 bits. The net gain is that DynaCT Micro is now visualizing 100-micron structures, and in clinical tests users have visualized small structures such as the tympani and membranes of the inner ear.
Also new is syngo DynaPBV Body, which takes the company's parenchymal blood volume (PBV) technology for brain imaging and adapts it for body work. DynaPBV enables physicians to visualize steady-state blood volume around tumors, helping them determine if an embolization was effective, for example.
Siemens is initially offering Artis Q.zen in a biplane configuration with two midsize 30 x 26-cm detectors. This configuration is most optimal for combination work and pediatric and electrophysiologic (EP) procedures. Artis Q.zen will have a red stripe on its detectors to differentiate the crystalline silicon technology from the amorphous silicon detectors used on Artis Q.
Two Artis Q.zen systems are installed in Europe and are acquiring images as part of clinical trials, Williams said. One site is doing body and neuro work while the other focuses on cardiology, with interventional and EP procedures.
Siemens plans to start shipping the Artis Q systems in Europe sometime in 2013, and a 510(k) is pending with the U.S. Food and Drug Administration. It aims to offer Artis Q.zen at a price point above its Artis zee angiography system, which will remain in the company's product line.