CHICAGO - New clinical applications in MRI, dose reduction programs in CT and interventional radiology, and continuing progress in its new women's health division are among the highlights in the RSNA booth of Philips Healthcare of Andover, MA, at this week's meeting.
MRI
In the MRI section of its booth, Philips is discussing applications for its Achieva 3.0T 3-tesla MRI scanner that optimize body and breast imaging. A particular highlight is the company's MultiTransmit multichannel radiofrequency transmission technology. First introduced as a work-in-progress at the 2008 RSNA meeting, Philips began shipping MultiTransmit in October, and expects to have 40 installations by the end of the year.
Philips believes that MultiTransmit will expand the clinical utility of 3-tesla MRI and make it suitable for mainstream clinical practice. Sites are reporting better image uniformity and consistency in image quality from patient to patient, regardless of patient size. MultiTransmit also results in a 40% improvement in speed -- for example, scans can be performed in five minutes on children with abdominal problems, which reduces the need for sedation.
Philips also has added a prostate imaging capability to its Elite suite of clinical applications. The package includes all the protocols needed to perform prostate studies, including spectroscopy, as well as the DynaCAD computer-aided detection (CAD) software.
In breast MRI, Philips is discussing its Elite package for breast MRI, and is touting clinical studies that indicate that it improves workflow by 30%. The package uses a 16-channel dedicated breast coil, and workflow is improved with a new MammoTrak trolley.
 |
| Sonalleve MR-HIFU is a high-intensity focused ultrasound package being shown as a work-in-progress. |
Sonalleve MR-HIFU is a new high-frequency focused ultrasound (HIFU) package that is being shown as a works-in-progress for noninvasive uterine fibroid treatment. When added to a Philips MRI scanner, the technology uses MRI guidance to direct focused ultrasound energy to shrink fibroids. The technology would require a premarket approval (PMA) application before it can be sold in the U.S., the company said.
CT
Dose reduction is an important topic at this year's meeting, and Philips is responding by unveiling iDose, the company's version of iterative reconstruction algorithms. Philips says that iDose can reduce the dose of coronary CT angiography studies to 0.25 to 1 mSv, a reduction of up to 80% over conventional filtered back projection reconstruction, with no loss of image quality, according to the company.
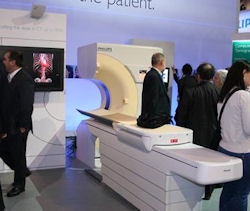 |
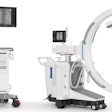
| This cut-in-half CT gantry is being used to highlight radiation dose awareness in the Philips booth. |
iDose will be available on the iCT and Brilliance 64 scanners, and will involve installing a new computer CPU and software. Philips expects deliveries to begin in the second or third quarter of 2010.
Philips is also discussing the development of "locked" protocols that make it more difficult for customer sites to change scanning protocols -- another response to concerns over radiation dose.
Women's imaging
Philips is demonstrating its growing commitment to women's imaging by highlighting its work-in-progress full-field digital mammography system, as well as a new breast imaging workstation.
MammoDiagnost DR is a flat-panel digital FFDM unit that is already shipping in Europe, and Philips is working on a PMA application for the system.
On the software side, Integral Breast Workspace is a new breast imaging workstation that includes viewing, diagnosis, and reporting functions, with reporting based on the American College of Radiology's BI-RADS lexicon. The workstation supports multimodality image review, automated display of prior images, and advanced applications like DynaCAD. Philips anticipates being able to ship the workstation in 2010.
Nuclear medicine
Philips is showcasing new clinical images from its BrightView XCT SPECT/CT unit, which now uses Astonish image reconstruction technology. This provides 50% better spatial resolution in bone and cardiac SPECT studies compared to SPECT studies using filtered back projection, according to the company.
BrightView XCT's hybrid imaging functionality positions it as a dual-purpose modality that can be a cost-effective replacement for a dedicated SPECT modality. In this situation, construction and room modification costs may be minimized because the system typically has the same footprint of the SPECT modality it is replacing.
Also being shown is the company's Gemini TF PET/CT system, designed to streamline the processes involved in staging, planning, therapy, and follow-up in oncology care.
Presented as a work-in-progress at the SNM 2009 meeting, Gemini TF is being shipped with Generation 3 time-of-flight software, which improves contrast resolution by 50%. Since June 2009, the company has received more than 100 orders for the Gemini TF, a fact that Philips attributes in part to being the fastest PET scanner it has manufactured. Data are captured in less than 500 picoseconds, without compromising image quality or attention to dose radiation efficiency.
Ultrasound
2009 is ultrasound's big year at Philips. Elastography, which uses conventional ultrasound to measure the compressibility and mechanical portion of a lesion, has been added to the company's iU22 premium ultrasound scanner. Approved and in use in Europe, elastography is being shown as a work-in-progress pending U.S. Food and Drug Administration clearance anticipated early in 2010.
The image guidance capabilities of the PercuNav instrument tracking system are also being demonstrated with the iU22 to show how the accuracy of needle biopsies and other interventional procedures may be improved. PercuNav displays the position, trajectory, and orientation of an instrument, working much like a global position system. It also offers the ability to synchronize stored images with real-time ultrasound using an image fusion function. CT images, for example, can be registered with corresponding ultrasound images even when there is respiratory motion.
PercuNav is commercially available in the United States, with availability anticipated by Philips in the first quarter of 2010 in the European Union and later in the year in Canada.
In addition to the expanded functionality of iU22, Philips is showcasing its portable premium ultrasound system, CX50 CompactXtreme. Designed like an oversized laptop computer, this mobile ultrasound system offers the features, functionality, and identical image quality as the cart-based iU22. It is equipped with Philips PureWave transducers, and supports the use of an integrated guidance tool. Targeted users include radiology applications in emergency departments, critical care, and intensive care units, and in women’s health centers, clinics, and mobile care vans.
In a special R&D section of the company's booth, Philips is demonstrating the use of shear-wave ultrasound elastography for liver fibrosis staging as a work-in-progress. A focused ultrasound pulse generates a shear wave that can be measured to determine the health of liver tissue as a means of detecting early signs of cirrhosis. The company expects to begin clinical trials in 2010.
X-ray
New x-ray developments in the Philips booth include DoseAware, a new program to help users of the company's interventional radiology systems better track radiation dose to patients and department staff.
 |
| Philips' DoseAware program uses dosimetry badges to generate radiation dose data wirelessly in real time. |
The company has developed dosimetry badges that provide the level of radiation dose a person in the interventional suite is receiving in real time. Data is sent wirelessly to a control panel, and staff can reduce their radiation exposure during procedures by repositioning themselves in the suite. DoseAware ships in January.
In addition, Philips is showing added support for multimodality imaging, with its 3D road mapping application enabling users of the company's angiography systems to bring 3D images from MRI and CT into the interventional suite to help guide procedures.
The firm is also showing a CX50 CompactXtreme scanner integrated with an Allura interventional lab to help guide procedures. The scanner can be operated from tableside to improve workflow. Philips is also now shipping the large-format FlexVison XL 8-megapixel in-room monitor shown as a work-in-progress at last year's meeting.
In digital radiography, the company is showing its DigitalDiagnost system with a new wireless detector for patients in wheelchairs and gurneys, as well as a swivel-top table that swings away from the overhead tube crane to make it easier to position patients. The wireless detector is scheduled to begin shipping in the next several months.
On its EasyDiagnost Eleva DRF digital radiography/fluoroscopy unit, Philips is showing a new configuration of the unit with a DR detector in a wall stand to make x-ray rooms more productive. The company is also showing Essenta DR Compact, a C-arm-based DR system. Philips estimates that the product will be 40% cheaper than a full DR suite; international shipments are already underway, and U.S. commercialization is expected in the first or second quarter of 2010.
Healthcare informatics
Making access to clinical information and images is the central focus of Philips healthcare informatics offerings, which include fully integrated RIS/PACS. Advanced clinical applications, including vessel exploration, virtual colonoscopy, and new orthopedic functionality, shown as works-in-progress last year, are now commercially available on its iSite diagnostic workstations.
For smaller institutions and emerging markets, Philips is also introducing a PACS configuration that it calls the Philips Medial Distribution Center. Modular and scalable, this offering provides easier access into iSite at a lower cost of ownership.
Customer services
With a weakened global economy, Philips Customer Services is emphasizing its support services for customers, ranging from country-specific diversified financing options through Philips Medical Capital, to utilization services that can help define modality needs, to flourishing sales of refurbished equipment.
Philips Medical Capital, a joint venture between Philips Healthcare and financing firm De Lage Landen, a division of Dutch banking giant Rabobank, offers a wide variety of creative financing solutions based on diverse needs of customers ranging from small private practices to mega-healthcare enterprises.
In the United States, a user pro forma analysis tool provides easy-to-develop financial models to assess cash flows and financing options of equipment. Similar software programs have been developed for countries throughout the world, based on country-specific financing regulations and preferred financial options.
New this year is Utilization Services, a remote monitoring service that acquires data about modality equipment utilization. The service helps its customers identify and minimize utilization inefficiencies, offering recommendations to improve procedural workflow and better optimize exam scheduling for modalities.
Some of the recommendations made to a customer may be for the acquisition of Diamond Selected refurbished systems, either to replace aging equipment or to supplement an installed base of diagnostic modalities to offer increased examination capacity at a lower price. In addition to parts replacement, all refurbished modalities contain the most current software upgrades available for the product, and where workstations are included, new computer hardware and display monitors, according to the company. All refurbishing work is performed in a new ISO 9000-certified manufacturing facility built in 2008 on the former Picker International campus in Cleveland.
The customer service division of Philips continues to expand its clinical CME training modules for radiologic technologists, as well as business-related courses for department administrators. Philips’ radiology specific training modules are certified by the American Society of Radiologic Technologists of Albuquerque, NM, and are potentially available for CME credit to more than 200,000 U.S. technologists alone.
By Brian Casey and Cynthia Keen
AuntMinnie.com staff writers
December 2, 2009
Related Reading
Philips partners with BrainLab, November 19, 2009
Philips SmartArc used for first time in U.S., November 13, 2009
Philips adds new apps for CX50 CompactXtreme, November 11, 2009
Philips completes Texas MRI install, November 10, 2009
Philips inks 8-year PACS/Xcelera deal, November 5, 2009
Copyright © 2009 AuntMinnie.com