CHICAGO - The latest generation of its dual-source CT technology, an MRI scanner optimized for breast imaging, and a work-in-progress digital breast tomosynthesis (DBT) technology are highlights for Siemens Healthcare at this year's RSNA show.
CT
 |
| Definition Flash is the latest generation of the company's dual-source CT technology. |
Siemens is emphasizing Definition Flash's ability to conduct CT scans at low radiation dose. The scanner was designed from the ground up to optimize radiation dose, according to company executives, and is able to conduct coronary CT angiography studies at a patient dose as low as 1 mSv. Definition Flash includes X-Care, a new dose-reduction technology that uses tube-side collimation to adapt the x-ray beam to the area being scanned, and turns it off in areas that aren't necessary for the image.
Siemens is also emphasizing the dual-energy scanning possibilities with the system. The company has developed a selective photon shield that that blocks unnecessary low-energy photons of the high-energy x-ray tube spectrum. This reduces patient radiation dose to the level of any standard single 120-kV scan, and it also enables the separation of high-energy and low-energy x-ray spectra during dual-energy imaging. Siemens has also developed a simplified protocol for conducting dual-energy studies that the company hopes will enable the technique to be used routinely.
In other CT news, Siemens is launching a new 20-slice version of its Somatom Definition AS single-source scanner, which previously was available in configurations from 40 to 128 slices. The new version will be targeted at budget-minded facilities but can be upgraded to 128 slices if the customer's needs change. The system will begin shipping in spring 2009.
MRI
Siemens is ramping up its activities in breast MRI with the launch of Magnetom Espree - Pink, a new version of the Espree 1.5-tesla scanner that's optimized for breast imaging. The scanner includes an 8-channel breast coil from Sentinelle Medical of Toronto that can be used for both imaging and biopsy applications.
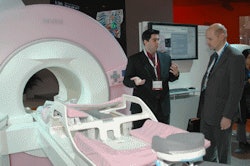 |
| Siemens designed Espree - Pink for breast MRI studies. |
Siemens is also talking up syngo Brevis, a multimodality workstation for breast MR and mammography, as well as new dedicated breast coils for 1.5-tesla and 3-tesla scanners using its Total image matrix (Tim) coil concept. An advanced imaging coil will be available with 16 radiofrequency channels.
Meanwhile, Siemens isn't ignoring men's health -- the company is highlighting syngo Tissue 4D, an application for analyzing dynamic data from prostate and liver MRI studies. Tissue 4D has two types of workflow, standard and pharmacokinetic modeling (PKM). PKM quantifies contrast uptake and can be used for oncology workflow and follow-up studies.
Another new MRI application, syngo TimCT Oncology, is designed to enable continuous table movement for extended field-of-view imaging. Siemens believes the application reduces the need for rescheduling multiple exams -- for example, a pelvic exam can be completed in as little as 20 minutes.
Finally, Siemens is introducing syngo Grace @ 3T, a quantitative MR breast spectroscopy application that monitors choline as a biomarker, providing information on the biochemical composition of breast lesions. Preliminary results indicate that detection of choline signal in breast tumors correlates with breast cancer, and Siemens believes that checking relative choline concentration during therapy makes treatment monitoring more reliable.
Mammography
Siemens is discussing its work-in-progress digital breast tomosynthesis technology. When commercially introduced, tomosynthesis will be available on the vendor's Mammomat Inspiration full-field digital mammography (FFDM) platform.
The vendor has also added stereotactic breast biopsy capability to Inspiration, allowing the unit to provide screening, diagnostics, biopsy, and eventually tomosynthesis on one platform.
The new biopsy unit slides on to the full-field detector of the mammography platform; the mammography system detects the biopsy unit and automatically switches all hardware and software configuration settings to the biopsy mode, Siemens said. Vertical or lateral needle access to lesions is available.
Inspiration, which is not yet available in the U.S., has been installed at more than 150 institutions worldwide, Siemens said.
In addition, the vendor is again displaying its NovationDR FFDM system. Novation features a 24 x 29-cm detector and allows for the integration of its Opdima digital spot imaging and digital biopsy system, Siemens said.
Siemens has also added new capabilities to its Mammotest prone breast biopsy table, including enhanced DICOM capabilities and stability functionality.
Other introductions include Siemens' syngo Multi-Modality Workplace for multimodality image viewing, as well as its MammoReport workflow engine. MammoReport provides image routing, blocking, and prefetching functionality.
PACS/Healthcare informatics
As was the case at the 2007 RSNA meeting, Siemens is spotlighting its syngo role-based portals this year. The portals, which include Portal Radiologist and the work-in-progress Portal Executive, Portal Referring Physician, and Portal Transcriptionist versions, provide individualized and context-based access to data and tools, according to the vendor.
Portal Executive is designed to allow executives and managers to proactively monitor department parameters in real-time, while Portal Transcriptionist helps transcriptionists achieve greater efficiency and accuracy when transcribing and correcting reports, Siemens said. The company said it has also enhanced Portal Radiologist and Portal Referring Physician.
Siemens is also emphasizing syngo Suite Essential, a package of preconfigured software, hardware, and services targeted at the U.S. diagnostic imaging center market. It's available on a fee-per-exam basis and is a modular offering, allowing providers to choose the options they want, according to the firm.
Because hardware and software are supported on a 24 x 7 basis by a Siemens support team, syngo Suite Essential doesn't require a dedicated onsite RIS/PACS administrator, according to the firm. Training and learning is provided via live, interactive online training, Siemens said.
In other developments, Siemens is debuting the latest release of its syngo Imaging XS PACS software. V70 includes new features for reading, planning exams, and reporting, as well as 3D visualization applications.
The firm's syngo Imaging PACS software has also received a new upgrade: V35 adds stability, scalability, and configurability enhancements. It also includes improved support functionality for customers transitioning from Siemens' legacy Magic PACS, handling of prior studies, and support of the vendor's syngo MammoReport and syngo Expert-i applications.
The new syngo Imaging release also feature a combination of computer-aided detection and advanced imaging capabilities within the PACS viewing workstation software, Siemens said.
In cardiac PACS developments, Siemens is pointing to version 7 of its syngo Dynamics offering. The new release features immediate data transfer between the company's Axiom Sensis XP hemodynamics monitoring system and syngo Dynamics reporting and image management system.
In addition, syngo Dynamics can incorporate data from GE Healthcare's MacLab v6.5 hemodynamic system. The release also adds a graphical reporting element to produce coronary tree diagrams and a new 17-segment wall-scoring model.
Siemens is also presenting the most recent version of its syngo Workflow RIS offering, including MLR V30C for markets outside the U.S. and SLR V30B for the U.S. market.
Ultrasound
In ultrasound developments, Siemens is highlighting its Acuson S2000 automated breast volume scanner (ABVS), which can automatically acquire full-field sonographic volumes of the breast. Features include an anatomical coronal plane and semiautomated reporting capabilities, Siemens said.
 |
| Acuson S2000 ABVS is designed to perform automated breast ultrasound. |
Siemens is also demonstrating its version 1.5 release of S2000. The latest release includes new applications and transducers, including its 14L5 SP small-parts transducer and 18L6 high-frequency transducer. 18L6 features a palm grip and sharper image detail capabilities, according to the vendor.
In works-in-progress highlights, Siemens is again discussing its silicon transducer research initiative. Development continues to move forward, and the company said it aims to commercialize silicon transducer technology in 2010.
The vendor also presented its work-in-progress ultrasound fusion technology, blending ultrasound and CT images.
X-ray
New applications for its x-ray portfolio are also a Siemens RSNA highlight. syngo iGuide Cappa is an electromagnetic needle guidance system that enables users to guide needle interventions without the use of radiation. The system accepts DICOM data from an interventional suite and can be used at tableside to reduce patient radiation dose during interventional procedures. The device has received FDA approval.
Being shown as a work-in-progress is syngo iFlow, which offers a color-coded view of digital subtraction angiography runs -- data are colorized depending on when in the exam the flow occurred.
Siemens is also emphasizing a new 56-inch 8-megapixel medical-grade digital display for its Artis zee-class angiography systems. The display is mounted on a ceiling-suspended arm and is designed to eliminate multiple individual displays that might be used in the interventional suite. Siemens has submitted a 510(k) application for the display, and the company estimates that it is about one year from commercial shipments.
Among work-in-progress developments for its zeego angiography systems are more fluid mechanical movements and a smoother look and feel of the system. Siemens is also taking orders from cardiac sites for the system.
Siemens has received 510(k) approval for the Ysio wireless digital radiography system, and the company is planning to install the first system in the U.S. at Central Baptist Hospital in Lexington, KY. The system sports a patient table with a 660-lb weight limit and is available in configurations with one or two digital detectors.
In other x-ray developments, Siemens introduced three new components for its Axiom Luminos dRF system, including a wireless detector, ceiling mounted x-ray tube, and new wall stand.
By Brian Casey and Erik L. Ridley
AuntMinnie.com staff writers
December 1, 2008
Related Reading
Siemens replaces Healthcare CEO, November 30, 2008
Siemens launches Definition Flash as flagship dual-source CT scanner, November 24, 2008
Siemens posts more revenue, less profit in Q4, November 13, 2008
Road to RSNA, Healthcare Informatics, Siemens Healthcare, November 10, 2008
Road to RSNA, Ultrasound, Siemens Healthcare, November 5, 2008
Copyright © 2008 AuntMinnie.com