CHICAGO - Dynamic perfusion CT imaging enables early assessment of the antivascular effects of antiangiogenic treatment in patients with advanced rectal cancer, researchers reported on Sunday at the 2008 RSNA annual meeting.
"CT perfusion acts as a surrogate end point to evaluate tumor response to antiangiogenic treatment," said Dr. Avinash Kambadakone Ramesh, a research fellow in the department of abdominal imaging and intervention at Massachusetts General Hospital (MGH) in Boston. RSNA presented Dr. Ramesh with the Research Trainee Award for his work.
"By looking at the initial tumor vasculature and the early changes, we can predict which patients will have a better outcome," added his mentor Dr. Dushyant Sahani, director of CT at MGH.
The study involved 25 patients with locally advanced rectal cancer and no distant metastases. Twenty-four of the patients had stage T3 disease, and one had stage 4 disease. The mean age of the patients, 19 of whom were men, was 56 years.
As initial treatment, 15 of the patients received bevacizumab alone, and 10 received bevacizumab, 5-fluorouracil, the epidermal growth factor receptor inhibitor erlotinib, and radiation.
CT perfusion, in which 70 mL of contrast media was injected at 7cc/sec and cine acquisition was performed for 45 seconds, was performed at baseline, 10 to 14 days after the start of treatment, and four weeks after completion of treatment. Data analysis was performed using CT Perfusion 3.0 software (GE Healthcare, Chalfont St. Giles, U.K.).
Standard perfusion parameters -- blood volume, blood flow, mean transit time, and permeability surface area product -- were compared before and after treatment.
By two weeks after treatment, blood flow and permeability surface area product had dropped significantly more in the bevacizumab monotherapy arm (43% and 28%, respectively), compared with the combination arm (24% and 8%).
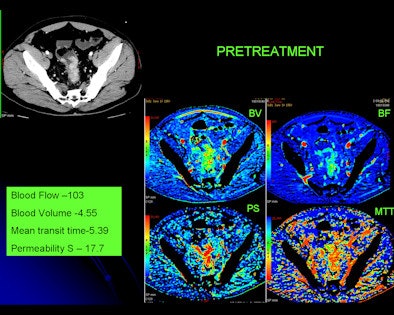 |
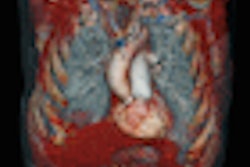
| Changes in tumor vasculature can be seen within days of starting antiangiogenic treatment. Images courtesy of Dr. Avinash Kambadakone Ramesh. |
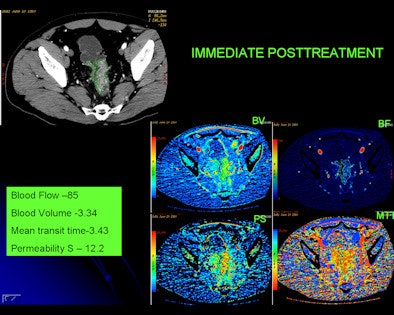 |
In actual numbers, blood flow dropped 87.6 mL/100g/min in the monotherapy arm, compared with 49.8 mL/100g/min in the combination arm. Permeability surface area product dropped 19.0 mL/100g/min in the monotherapy arm, versus 13.6 mL/100g/min in the combination arm.
After completion of treatment, tumors of patients in the monotherapy arm showed a significantly greater reduction in blood flow of 74.5 mL/100g/min, compared with 18.8 mL/100g/min in the combination arm.
"There were tumor perfusion changes in both groups, but they were considerably more substantial in the bevacizumab group," Ramesh said.
Sixteen of the 15 patients had a long-term follow-up of more than 24 months. Three of the patients had an unfavorable prognosis with the occurrence of metastases.
The patients with an unfavorable prognosis had higher baseline blood flow values than those with a favorable prognosis (87 versus 72 mL/100g/min).
Patients who had a favorable outcome showed a significantly greater decrease in blood flow (81% versus 68%), blood volume (52% versus 1%), and permeability surface area product (54% versus 23%) after completion of treatment, compared with patients with an unfavorable outcome.
"Since tumor vasculature is affected early on, we can measure the treatment effect within days of initiating treatment" Sahani said. "It's not like tumor size and other traditional measures of treatment response, which take weeks or months to measure."
What is not yet known, he said, is "whether we should change treatment based on early vasculature changes. That's something we need to find out."
By Charlene Laino
AuntMinnie.com contributing writer
December 1, 2008
Related Reading
CT perfusion scan spots acute stroke in the emergency setting, November 5, 2008
Study tests low dose ranges for perfusion CT, December 10, 2007
256-slice perfusion CT equivalent to SPECT for arteriosclerosis detection, November 13, 2007
ASPECTS stroke scoring improved with perfusion CT, February 21, 2007
Distinct CT perfusion parameters help assess HCC, November 29, 2005
Copyright © 2008 AuntMinnie.com