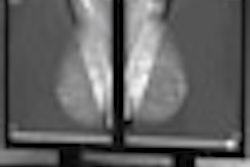
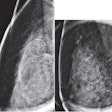
CHICAGO - MRI proves better than mammography for measuring breast tumor response to neoadjuvant chemotherapy, according to research performed at the University of California, San Francisco (UCSF) and eight other U.S. academic institutions for the American College of Radiology Imaging Network (ACRIN) 6657 study.
Nola Hylton, Ph.D., a principal investigator from UCSF, presented the study results at the RSNA meeting. The study is being conducted as part of the national I-SPY (Investigation of Serial Studies to Predict Your Therapeutic Response With Imaging and Molecular Analysis) trial of imaging and biomarkers for monitoring and guiding preoperative treatment for women with breast cancer. The collaboration is supported by the Cancer and Leukemia Group B Foundation (CALGB), ACRIN, and the National Cancer Institute's InterSPORE program and its Center for Bioinformatics.
Between May 2002 and March 2006, the study enrolled 216 women with 3 cm or greater invasive breast cancer who had received an anthracycline-cyclophosphamide neoadjuvant chemotherapy regimen followed optionally by a taxane.
"Our study had two goals: to evaluate the ability of MRI to predict three-year disease free survival, and to evaluate whether MRIs taken early in treatment can predict a patient's response to treatment," Hylton said.
The women were scanned four times: once before therapy, once after the first cycle of chemotherapy, between the anthracycline and Taxol agents, and finally prior to surgery, to detect residual disease and evaluate the post-treatment sensitivity and specificity of MRI. Compliance was high, with 97% of the women returning for the early treatment scan, 93% for the between-agents scan, and 97% for the presurgery scan.
The study took four measurements with MRI, including the longest diameter of the tumor, its morphologic pattern and volume, and the signal enhancement ratio (SER), according to Hylton.
After surgery, 43% of the patients were complete responders, 38% were partial responders, 10% demonstrated stable disease, and 3% had progressive disease; the remainder was not measured. In terms of pathologic response, 38% of patients demonstrated pathologically complete response, while 58% demonstrated nonpathologically complete response. Seventy-one percent of the lesions were mass type and 23% were regional cancers.
The team found that in the comparison of clinical, mammographic, and MRI measures of tumor size, MRI tumor volume was most predictive of pathologic residual disease size and clinical response.
"We found that after neoadjuvant chemotherapy, MRI is more accurate than clinical exam or mammography in estimating residual disease size and predicting response to treatment," Hylton said. "Mammography does not appear to add value in this setting.
The team continues to follow patients to collect three-year disease free survival data and expects that dataset to be complete in June 2009.
By Kate Madden Yee
AuntMinnie.com staff writer
November 30, 2008
Related Reading
MRI beats mammo for gauging breast cancer treatment response, October 7, 2008
MRI techniques show early tumor response to radiation, September 30, 2008
Breast MRI can guide radiation therapy to lymph nodes, September 22, 2008
Routine MRI at breast cancer diagnosis linked to treatment delays, September 9, 2008
Breast MRI gains momentum for screening high-risk women, September 2, 2008
Copyright © 2008 AuntMinnie.com